Peptide Information
General Information of This Peptide
| Peptide ID |
PEP00105
|
|||||
|---|---|---|---|---|---|---|
| Peptide Name |
Octreotide
|
|||||
| Structure |
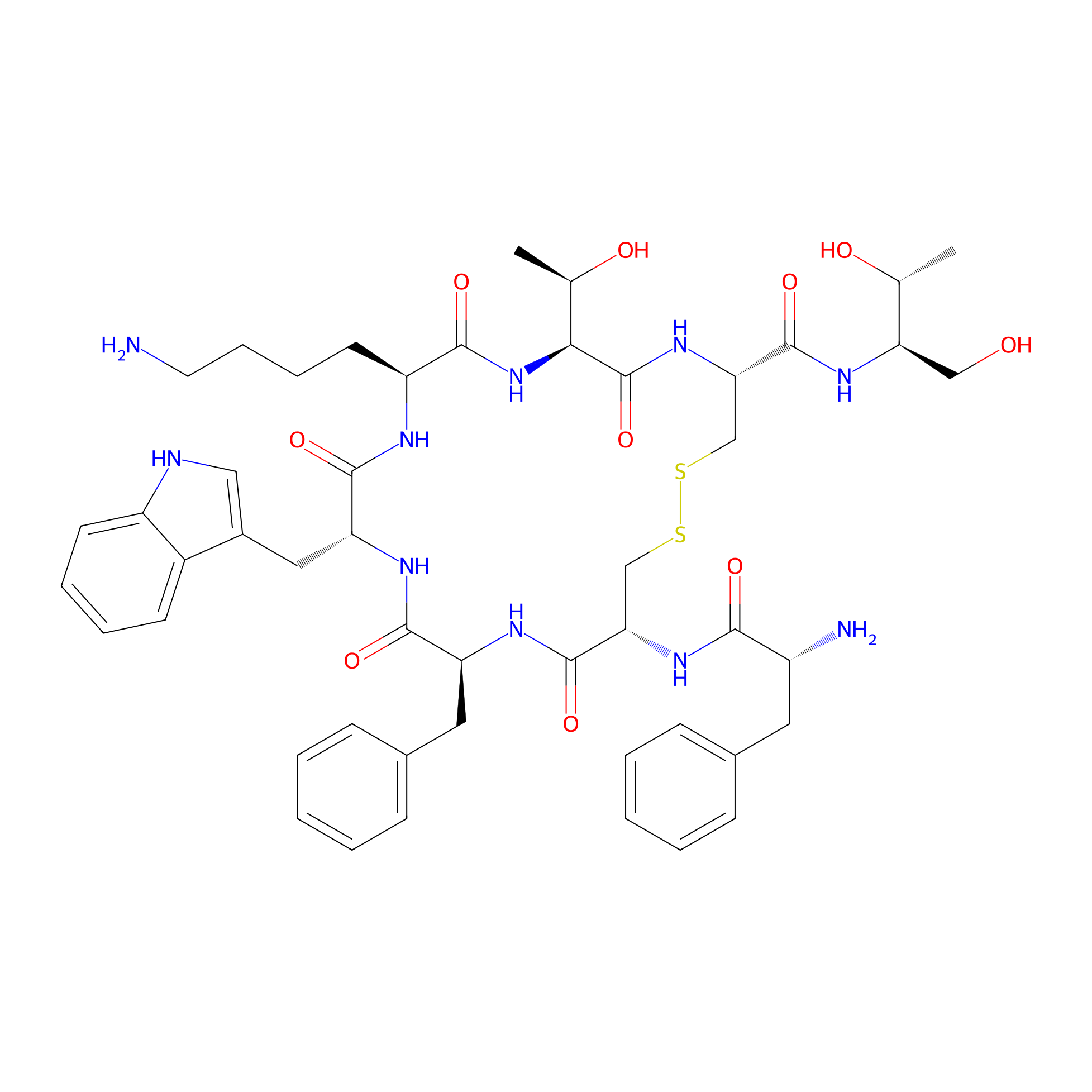
|
|||||
| Sequence |
NH2-FCFWKTCT
|
|||||
| Peptide Type |
Cyclic
|
|||||
| Receptor Name |
Somatostatin receptor type 1 (SSTR1)
|
Receptor Info | ||||
| PDC Transmembrane Types | Cell targeting peptides (CTPs) | |||||
| Formula |
C49H66N10O10S2
|
|||||
| Isosmiles |
C[C@@H](O)[C@@H]1NC(=O)[C@H](CCCCN)NC(=O)[C@@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@@H](NC(=O)[C@H](N)Cc2ccccc2)CSSC[C@@H](C(=O)N[C@H](CO)[C@@H](C)O)NC1=O
|
|||||
| InChI |
InChI=1S/C49H66N10O10S2/c1-28(61)39(25-60)56-48(68)41-27-71-70-26-40(57-43(63)34(51)21-30-13-5-3-6-14-30)47(67)54-37(22-31-15-7-4-8-16-31)45(65)55-38(23-32-24-52-35-18-10-9-17-33(32)35)46(66)53-36(19-11-12-20-50)44(64)59-42(29(2)62)49(69)58-41/h3-10,13-18,24,28-29,34,36-42,52,60-62H,11-12,19-23,25-27,50-51H2,1-2H3,(H,53,66)(H,54,67)(H,55,65)(H,56,68)(H,57,63)(H,58,69)(H,59,64)/t28-,29-,34-,36+,37+,38-,39-,40+,41+,42+/m1/s1
|
|||||
| InChIKey |
DEQANNDTNATYII-OULOTJBUSA-N
|
|||||
| Pharmaceutical Properties |
Molecule Weight
|
1019.261
|
Polar area
|
332.22
|
||
|
Complexity
|
1018.44048
|
xlogp Value
|
-0.8054
|
|||
|
Heavy Count
|
71
|
Rot Bonds
|
17
|
|||
|
Hbond acc
|
14
|
Hbond Donor
|
13
|
|||
The Activity Data of This Peptide
| Peptide Activity Information 1 | [1], [2] | |||||
| IC50 | > 1000 nmol/L | |||||
|---|---|---|---|---|---|---|
| Binding Affinity Assay |
CHO-K1 and CCL39 cells stably expressing human sst1-5 (hsst1-5) were grown as described previously . All culture reagents were supplied by GIBCO/BRL and Life Technologies (Grand Island, NY). Cell membrane pellets were prepared and receptor autoradiography was performed on pellet sections (mounted on microscope slides), as described in detail previously . For each of the tested compounds, complete displacement experiments were performed with the universal somatostatin radioligand [125I]-[Leu8,D-Trp22,Trp25]-somatostatin-28 using increasing concentrations of the MetalloIII-DOTA-peptide ranging from 0.1 to 1,000nmol/l. Somatostatin-28 was run in parallel as control using the same increasing concentrations. IC50values were calculated after quantification of the data using a computer-assisted image processing system. Tissue standards (autoradiographic [125I] microscales, Amersham, UK) containing known amounts of isotopes, cross-calibrated to tissue-equivalent ligand concentrations, were used for quantification . The concentrations of the peptide solutions were measured by UV spectroscopy (NOC-ATE, 280nm = 9,855cm-1mol-1dm3, BOC-ATE, 280nm = 7,570cm-1mol-1dm3, TOC, 280nm> = 6,849cm-1mol-1dm3, NOC,280nm> = 9,850cm-1mol-1dm3).
Click to Show/Hide
|
|||||
| Experimental Condition | CHO-K1 and CCL39 cellsSSRT3 | |||||
| Peptide Activity Information 2 | [1], [2] | |||||
| IC50 | > 10000 nmol/L | |||||
| Binding Affinity Assay |
CHO-K1 and CCL39 cells stably expressing human sst1-5 (hsst1-5) were grown as described previously . All culture reagents were supplied by GIBCO/BRL and Life Technologies (Grand Island, NY). Cell membrane pellets were prepared and receptor autoradiography was performed on pellet sections (mounted on microscope slides), as described in detail previously . For each of the tested compounds, complete displacement experiments were performed with the universal somatostatin radioligand [125I]-[Leu8,D-Trp22,Trp25]-somatostatin-28 using increasing concentrations of the MetalloIII-DOTA-peptide ranging from 0.1 to 1,000nmol/l. Somatostatin-28 was run in parallel as control using the same increasing concentrations. IC50values were calculated after quantification of the data using a computer-assisted image processing system. Tissue standards (autoradiographic [125I] microscales, Amersham, UK) containing known amounts of isotopes, cross-calibrated to tissue-equivalent ligand concentrations, were used for quantification . The concentrations of the peptide solutions were measured by UV spectroscopy (NOC-ATE, 280nm = 9,855cm-1mol-1dm3, BOC-ATE, 280nm = 7,570cm-1mol-1dm3, TOC, 280nm> = 6,849cm-1mol-1dm3, NOC,280nm> = 9,850cm-1mol-1dm3).
Click to Show/Hide
|
|||||
| Experimental Condition | CHO-K1 and CCL39 cellsSSRT1 | |||||
| Peptide Activity Information 3 | [1], [2] | |||||
| IC50 | 0.20 ± 0.04 nmol/L | |||||
| Binding Affinity Assay |
CHO-K1 and CCL39 cells stably expressing human sst1-5 (hsst1-5) were grown as described previously . All culture reagents were supplied by GIBCO/BRL and Life Technologies (Grand Island, NY). Cell membrane pellets were prepared and receptor autoradiography was performed on pellet sections (mounted on microscope slides), as described in detail previously . For each of the tested compounds, complete displacement experiments were performed with the universal somatostatin radioligand [125I]-[Leu8,D-Trp22,Trp25]-somatostatin-28 using increasing concentrations of the MetalloIII-DOTA-peptide ranging from 0.1 to 1,000nmol/l. Somatostatin-28 was run in parallel as control using the same increasing concentrations. IC50values were calculated after quantification of the data using a computer-assisted image processing system. Tissue standards (autoradiographic [125I] microscales, Amersham, UK) containing known amounts of isotopes, cross-calibrated to tissue-equivalent ligand concentrations, were used for quantification . The concentrations of the peptide solutions were measured by UV spectroscopy (NOC-ATE, 280nm = 9,855cm-1mol-1dm3, BOC-ATE, 280nm = 7,570cm-1mol-1dm3, TOC, 280nm> = 6,849cm-1mol-1dm3, NOC,280nm> = 9,850cm-1mol-1dm3).
Click to Show/Hide
|
|||||
| Experimental Condition | CHO-K1 and CCL39 cellsSSRT2 | |||||
| Peptide Activity Information 4 | [1], [2] | |||||
| IC50 | 1.6 ± 0.4 nmol/L | |||||
| Binding Affinity Assay |
CHO-K1 and CCL39 cells stably expressing human sst1-5 (hsst1-5) were grown as described previously . All culture reagents were supplied by GIBCO/BRL and Life Technologies (Grand Island, NY). Cell membrane pellets were prepared and receptor autoradiography was performed on pellet sections (mounted on microscope slides), as described in detail previously . For each of the tested compounds, complete displacement experiments were performed with the universal somatostatin radioligand [125I]-[Leu8,D-Trp22,Trp25]-somatostatin-28 using increasing concentrations of the MetalloIII-DOTA-peptide ranging from 0.1 to 1,000nmol/l. Somatostatin-28 was run in parallel as control using the same increasing concentrations. IC50values were calculated after quantification of the data using a computer-assisted image processing system. Tissue standards (autoradiographic [125I] microscales, Amersham, UK) containing known amounts of isotopes, cross-calibrated to tissue-equivalent ligand concentrations, were used for quantification . The concentrations of the peptide solutions were measured by UV spectroscopy (NOC-ATE, 280nm = 9,855cm-1mol-1dm3, BOC-ATE, 280nm = 7,570cm-1mol-1dm3, TOC, 280nm> = 6,849cm-1mol-1dm3, NOC,280nm> = 9,850cm-1mol-1dm3).
Click to Show/Hide
|
|||||
| Experimental Condition | CHO-K1 and CCL39 cellsSSRT2 | |||||
| Peptide Activity Information 5 | [1], [2] | |||||
| IC50 | 187 ± 50 nmol/L | |||||
| Binding Affinity Assay |
CHO-K1 and CCL39 cells stably expressing human sst1-5 (hsst1-5) were grown as described previously . All culture reagents were supplied by GIBCO/BRL and Life Technologies (Grand Island, NY). Cell membrane pellets were prepared and receptor autoradiography was performed on pellet sections (mounted on microscope slides), as described in detail previously . For each of the tested compounds, complete displacement experiments were performed with the universal somatostatin radioligand [125I]-[Leu8,D-Trp22,Trp25]-somatostatin-28 using increasing concentrations of the MetalloIII-DOTA-peptide ranging from 0.1 to 1,000nmol/l. Somatostatin-28 was run in parallel as control using the same increasing concentrations. IC50values were calculated after quantification of the data using a computer-assisted image processing system. Tissue standards (autoradiographic [125I] microscales, Amersham, UK) containing known amounts of isotopes, cross-calibrated to tissue-equivalent ligand concentrations, were used for quantification . The concentrations of the peptide solutions were measured by UV spectroscopy (NOC-ATE, 280nm = 9,855cm-1mol-1dm3, BOC-ATE, 280nm = 7,570cm-1mol-1dm3, TOC, 280nm> = 6,849cm-1mol-1dm3, NOC,280nm> = 9,850cm-1mol-1dm3).
Click to Show/Hide
|
|||||
| Experimental Condition | CHO-K1 and CCL39 cellsSSRT5 | |||||
| Peptide Activity Information 6 | [1], [2] | |||||
| IC50 | 300 ± 140 nmol/L | |||||
| Binding Affinity Assay |
CHO-K1 and CCL39 cells stably expressing human sst1-5 (hsst1-5) were grown as described previously . All culture reagents were supplied by GIBCO/BRL and Life Technologies (Grand Island, NY). Cell membrane pellets were prepared and receptor autoradiography was performed on pellet sections (mounted on microscope slides), as described in detail previously . For each of the tested compounds, complete displacement experiments were performed with the universal somatostatin radioligand [125I]-[Leu8,D-Trp22,Trp25]-somatostatin-28 using increasing concentrations of the MetalloIII-DOTA-peptide ranging from 0.1 to 1,000nmol/l. Somatostatin-28 was run in parallel as control using the same increasing concentrations. IC50values were calculated after quantification of the data using a computer-assisted image processing system. Tissue standards (autoradiographic [125I] microscales, Amersham, UK) containing known amounts of isotopes, cross-calibrated to tissue-equivalent ligand concentrations, were used for quantification . The concentrations of the peptide solutions were measured by UV spectroscopy (NOC-ATE, 280nm = 9,855cm-1mol-1dm3, BOC-ATE, 280nm = 7,570cm-1mol-1dm3, TOC, 280nm> = 6,849cm-1mol-1dm3, NOC,280nm> = 9,850cm-1mol-1dm3).
Click to Show/Hide
|
|||||
| Experimental Condition | CHO-K1 and CCL39 cellsSSRT4 | |||||
| Peptide Activity Information 7 | [1], [2] | |||||
| IC50 | 377 ± 18 nmol/L | |||||
| Binding Affinity Assay |
CHO-K1 and CCL39 cells stably expressing human sst1-5 (hsst1-5) were grown as described previously . All culture reagents were supplied by GIBCO/BRL and Life Technologies (Grand Island, NY). Cell membrane pellets were prepared and receptor autoradiography was performed on pellet sections (mounted on microscope slides), as described in detail previously . For each of the tested compounds, complete displacement experiments were performed with the universal somatostatin radioligand [125I]-[Leu8,D-Trp22,Trp25]-somatostatin-28 using increasing concentrations of the MetalloIII-DOTA-peptide ranging from 0.1 to 1,000nmol/l. Somatostatin-28 was run in parallel as control using the same increasing concentrations. IC50values were calculated after quantification of the data using a computer-assisted image processing system. Tissue standards (autoradiographic [125I] microscales, Amersham, UK) containing known amounts of isotopes, cross-calibrated to tissue-equivalent ligand concentrations, were used for quantification . The concentrations of the peptide solutions were measured by UV spectroscopy (NOC-ATE, 280nm = 9,855cm-1mol-1dm3, BOC-ATE, 280nm = 7,570cm-1mol-1dm3, TOC, 280nm> = 6,849cm-1mol-1dm3, NOC,280nm> = 9,850cm-1mol-1dm3).
Click to Show/Hide
|
|||||
| Experimental Condition | CHO-K1 and CCL39 cellsSSRT5 | |||||
| Peptide Activity Information 8 | [1], [2] | |||||
| IC50 | 523 ± 239 nmol/L | |||||
| Binding Affinity Assay |
CHO-K1 and CCL39 cells stably expressing human sst1-5 (hsst1-5) were grown as described previously . All culture reagents were supplied by GIBCO/BRL and Life Technologies (Grand Island, NY). Cell membrane pellets were prepared and receptor autoradiography was performed on pellet sections (mounted on microscope slides), as described in detail previously . For each of the tested compounds, complete displacement experiments were performed with the universal somatostatin radioligand [125I]-[Leu8,D-Trp22,Trp25]-somatostatin-28 using increasing concentrations of the MetalloIII-DOTA-peptide ranging from 0.1 to 1,000nmol/l. Somatostatin-28 was run in parallel as control using the same increasing concentrations. IC50values were calculated after quantification of the data using a computer-assisted image processing system. Tissue standards (autoradiographic [125I] microscales, Amersham, UK) containing known amounts of isotopes, cross-calibrated to tissue-equivalent ligand concentrations, were used for quantification . The concentrations of the peptide solutions were measured by UV spectroscopy (NOC-ATE, 280nm = 9,855cm-1mol-1dm3, BOC-ATE, 280nm = 7,570cm-1mol-1dm3, TOC, 280nm> = 6,849cm-1mol-1dm3, NOC,280nm> = 9,850cm-1mol-1dm3).
Click to Show/Hide
|
|||||
| Experimental Condition | CHO-K1 and CCL39 cellsSSRT4 | |||||
Each Peptide-drug Conjugate Related to This Peptide
Full Information of The Activity Data of The PDC(s) Related to This Peptide
111In-DTPA-octreotide [Approved]
Obtained from the Model Organism Data
| Experiment 1 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Digestive system neoplasms | ||||
| Efficacy Data | Tumor cell uptake rate |
47%
|
|||
| Administration Time | 4 h | ||||
| Administration Dosage | 10 nM | ||||
| MOA of PDC |
In our previous study, d-Phe at position 1 in 111In-DTPA-d-Phe1-octreotide was replaced with Asp to minimize the change in molecular size. While the Phe3D-Trp4Lys5Thr6 sequence in octreotide is important for binding to the somatostatin receptor, the majority of octreotide derivatives with high somatostatin receptor affinity possess an N-terminal amino acid with an aromatic side chain, such as d-Phe or naphthylalanine. With these findings in mind, we designed, synthesized and evaluated not only the previously reported 111In-DTPA-Asp1-octreotide but also 111In-DTPA-Asp0-d-Phe1-octreotide, in which an Asp was incorporated between DTPA and N-terminal d-Phe in 111In-DTPA-d-Phe1-octreotide.
Click to Show/Hide
|
||||
| Description |
Figure 3 shows the results of cellular uptake experiments. When incubated with111In-DTPA-Asp1-octreotide, the uptake levels of radioactivity by AR42J cells were extremely low compared with those for111In-DTPA-d-Phe1-octreotide. In contrast, the uptake levels of111In-DTPA-Asp0-d-Phe1-octreotide increased in a time-dependent manner. The uptake levels of111In-DTPA-Asp0-d-Phe1-octreotide were not significantly different from those for111In-DTPA-d-Phe1-octreotide at 4h. For 111In-DTPA-Asp0-d-Phe1-octreotide, cell uptake experiments were conducted in the presence of varying concentrations of an unmodified octreotide. In Figure 4, uptake levels are shown as a percentage of the radioactivity level in the absence of the unmodified octreotide. The uptake levels of 111In-DTPA-Asp0-d-Phe1-octreotide were reduced in association with the concentration of the unmodified octreotide. The radioactivity uptake level was reduced by approximately 90% at 1 M concentration of the octreotide.
Click to Show/Hide
|
||||
| In Vivo Model | AR42J tumor-bearing mice model. | ||||
[18F]AlF-NOTA-octreotide [Investigative]
Identified from the Human Clinical Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor-induced osteomalacia | ||||
| Efficacy Data | Specificity |
100%
|
|||
| Patients Enrolled |
17 patients with hypophosphatemic osteomalacia suspected to be TIO.
|
||||
| Administration Dosage | 3.7 MBq (0.10 mCi) per kilogram of body weight | ||||
| Evaluation Method | 18F-OC PET/CT assay | ||||
| MOA of PDC |
18F-OC PET/CT scan is useful in the detection of tumors causing TIO. Further studies with larger patient populations are needed to validate the result.
|
||||
| Description |
The 18F-OC PET/CT scans were positive in 14 patients. Furthermore, 4 of 14 patients were scanned with both 18F-OC and 68Ga-DOTATATE PET/CT. Both studies were able to localize the tumor in all 4 patients. In total, 14 patients had surgery to remove the lesions. Postsurgical pathological examination confirmed causative tumors in these patients, whose symptoms diminished promptly. Serum phosphate levels normalized, confirming the diagnosis of TIO. 18F-OC PET/CT sensitivity, specificity, and accuracy were 87.5%, 100%, and 88.2% respectively. 18F-OC PET/CT findings affected patient management in 88.2% of cases.
Click to Show/Hide
|
||||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor-induced osteomalacia | ||||
| Efficacy Data | Sensitivity |
87.50%
|
|||
| Patients Enrolled |
17 patients with hypophosphatemic osteomalacia suspected to be TIO.
|
||||
| Administration Dosage | 3.7 MBq (0.10 mCi) per kilogram of body weight | ||||
| Evaluation Method | 18F-OC PET/CT assay | ||||
| MOA of PDC |
18F-OC PET/CT scan is useful in the detection of tumors causing TIO. Further studies with larger patient populations are needed to validate the result.
|
||||
| Description |
The 18F-OC PET/CT scans were positive in 14 patients. Furthermore, 4 of 14 patients were scanned with both 18F-OC and 68Ga-DOTATATE PET/CT. Both studies were able to localize the tumor in all 4 patients. In total, 14 patients had surgery to remove the lesions. Postsurgical pathological examination confirmed causative tumors in these patients, whose symptoms diminished promptly. Serum phosphate levels normalized, confirming the diagnosis of TIO. 18F-OC PET/CT sensitivity, specificity, and accuracy were 87.5%, 100%, and 88.2% respectively. 18F-OC PET/CT findings affected patient management in 88.2% of cases.
Click to Show/Hide
|
||||
| Experiment 3 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Neuroendocrine tumour | ||||
| Efficacy Data | Median detection ratio |
91.10%
|
|||
| Patients Enrolled |
75 patients with histologically confirmed neuroendocrine tumours and routine clinical.
|
||||
| MOA of PDC |
18F-AlF-OC is noninferior and even superior to 68Ga-DOTATATE/NOC PET in NET patients. This validates 18F-AlF-OC as an option for clinical practice somatostatin receptor PET.
|
||||
| Description |
In total, 4,709 different tumor lesions were detected: 3,454 with 68Ga-DOTATATE/NOC and 4,278 with 18F-AlF-OC. The mean DR with 18F-AlF-OC was significantly higher than with 68Ga-DOTATATE/NOC (91.1% vs. 75.3%; P < 10-5).
|
||||
| Experiment 4 Reporting the Activity Data of This PDC | [4] | ||||
| Indication | Neuroendocrine tumour | ||||
| Efficacy Data | Difference in detection ratio |
15.80%
|
|||
| Patients Enrolled |
75 patients with histologically confirmed neuroendocrine tumours and routine clinical.
|
||||
| MOA of PDC |
18F-AlF-OC is noninferior and even superior to 68Ga-DOTATATE/NOC PET in NET patients. This validates 18F-AlF-OC as an option for clinical practice somatostatin receptor PET.
|
||||
| Description |
The resulting mean DDR was 15.8%, with a lower margin of the 95% CI (95% CI, 9.6%-22.0%) higher than -15%, which is the prespecified boundary for noninferiority. The mean DDRs for the 68Ga-DOTATATE and 68Ga-DOTANOC subgroups were 11.8% (95% CI, 4.3-19.3) and 27.5% (95% CI, 17.8-37.1), respectively.
|
||||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor-induced osteomalacia | ||||
| Efficacy Data | Accuracy |
88.20%
|
|||
| Patients Enrolled |
17 patients with hypophosphatemic osteomalacia suspected to be TIO.
|
||||
| Administration Dosage | 3.7 MBq (0.10 mCi) per kilogram of body weight | ||||
| Evaluation Method | 18F-OC PET/CT assay | ||||
| MOA of PDC |
18F-OC PET/CT scan is useful in the detection of tumors causing TIO. Further studies with larger patient populations are needed to validate the result.
|
||||
| Description |
The 18F-OC PET/CT scans were positive in 14 patients. Furthermore, 4 of 14 patients were scanned with both 18F-OC and 68Ga-DOTATATE PET/CT. Both studies were able to localize the tumor in all 4 patients. In total, 14 patients had surgery to remove the lesions. Postsurgical pathological examination confirmed causative tumors in these patients, whose symptoms diminished promptly. Serum phosphate levels normalized, confirming the diagnosis of TIO. 18F-OC PET/CT sensitivity, specificity, and accuracy were 87.5%, 100%, and 88.2% respectively. 18F-OC PET/CT findings affected patient management in 88.2% of cases.
Click to Show/Hide
|
||||
Octreotide doxorubicin conjugate [Investigative]
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [5] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
27.14 ± 2.47 μM
|
|||
| Administration Time | 72 h | ||||
| Evaluation Method | CellTiter-Glo cell proliferation assay | ||||
| MOA of PDC |
In the present work, we introduce a new approach to overcome all the aforementioned limitations. The cytotoxic drug doxorubicin is coupled to the tumor-targeting vector octreotide via a disulfide-intercalating cross-linking reagent. On the one hand, this reagent creates an oxime bond with the drug, and on the other hand, two disulfides with octreotide to keep the cyclic structure of the peptide. The combination of a hydrolytically stable oxime bond and disulfides leads to the formation of a novel bioconjugate superior to any previous anticancer drug-somatostatin analog hybrid as it allows the efficient release of the toxic cargo within the reducing environment of cancer cells. The versatility of the linker molecule described here will enable its future application not only in targeted drug delivery, but also in the chemical modification of therapeutic proteins.
Click to Show/Hide
|
||||
| Description |
We selected cells, where doxorubicin is typically applied and which overexpress somatostatin receptors, like the human pancreatic carcinoma cell line MIA PaCa-2 or MCF-7. Doxorubicins antiproliferative action on MIA PaCa-2 after 72 h incubation with the drug was as expected very strong (IC50 = 0.80 ± 0.13 μM), whereas octreotide was not able to inhibit cell growth by 50% up to a concentration of 150 μM. The cytotoxic effects of the conjugate expressed, as half maximal inhibitory concentration was much stronger compared to the precursor peptide, but, nevertheless, lower than that of doxorubicin (IC50 = 31.50 ± 1.74 μM). This characteristic feature of 12 is attributed to the different cellular uptake mechanisms of the substances. Doxorubicin is taken up quickly by passive diffusion, while octreotide enters cells by receptor-mediated endocytosis. Furthermore, it needs to be considered that the conjugate releases after cleavage by glutathione a doxorubicin derivative, which is still carrying a small cross-linker residue. Previous studies have shown that these types of molecules are, nevertheless, capable of successfully interacting with DNA, to mediate their cytotoxic properties, but to a lesser extent.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [5] | ||||
| Indication | Pancreatic carcinoma | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
31.50 ± 1.74 μM
|
|||
| Administration Time | 72 h | ||||
| Evaluation Method | CellTiter-Glo cell proliferation assay | ||||
| MOA of PDC |
In the present work, we introduce a new approach to overcome all the aforementioned limitations. The cytotoxic drug doxorubicin is coupled to the tumor-targeting vector octreotide via a disulfide-intercalating cross-linking reagent. On the one hand, this reagent creates an oxime bond with the drug, and on the other hand, two disulfides with octreotide to keep the cyclic structure of the peptide. The combination of a hydrolytically stable oxime bond and disulfides leads to the formation of a novel bioconjugate superior to any previous anticancer drug-somatostatin analog hybrid as it allows the efficient release of the toxic cargo within the reducing environment of cancer cells. The versatility of the linker molecule described here will enable its future application not only in targeted drug delivery, but also in the chemical modification of therapeutic proteins.
Click to Show/Hide
|
||||
| Description |
We selected cells, where doxorubicin is typically applied and which overexpress somatostatin receptors, like the human pancreatic carcinoma cell line MIA PaCa-2 or MCF-7. Doxorubicins antiproliferative action on MIA PaCa-2 after 72 h incubation with the drug was as expected very strong (IC50 = 0.80 ± 0.13 μM), whereas octreotide was not able to inhibit cell growth by 50% up to a concentration of 150 μM. The cytotoxic effects of the conjugate expressed, as half maximal inhibitory concentration was much stronger compared to the precursor peptide, but, nevertheless, lower than that of doxorubicin (IC50 = 31.50 ± 1.74 μM). This characteristic feature of 12 is attributed to the different cellular uptake mechanisms of the substances. Doxorubicin is taken up quickly by passive diffusion, while octreotide enters cells by receptor-mediated endocytosis. Furthermore, it needs to be considered that the conjugate releases after cleavage by glutathione a doxorubicin derivative, which is still carrying a small cross-linker residue. Previous studies have shown that these types of molecules are, nevertheless, capable of successfully interacting with DNA, to mediate their cytotoxic properties, but to a lesser extent.
Click to Show/Hide
|
||||
| In Vitro Model | Pancreatic ductal adenocarcinoma | MIA PaCa-2 cell | CVCL_0428 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [5] | ||||
| Indication | Pancreatic adenocarcinoma | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
48.90 ± 5.40 μM
|
|||
| Administration Time | 72 h | ||||
| Evaluation Method | CellTiter-Glo cell proliferation assay | ||||
| MOA of PDC |
In the present work, we introduce a new approach to overcome all the aforementioned limitations. The cytotoxic drug doxorubicin is coupled to the tumor-targeting vector octreotide via a disulfide-intercalating cross-linking reagent. On the one hand, this reagent creates an oxime bond with the drug, and on the other hand, two disulfides with octreotide to keep the cyclic structure of the peptide. The combination of a hydrolytically stable oxime bond and disulfides leads to the formation of a novel bioconjugate superior to any previous anticancer drug-somatostatin analog hybrid as it allows the efficient release of the toxic cargo within the reducing environment of cancer cells. The versatility of the linker molecule described here will enable its future application not only in targeted drug delivery, but also in the chemical modification of therapeutic proteins.
Click to Show/Hide
|
||||
| Description |
We selected cells, where doxorubicin is typically applied and which overexpress somatostatin receptors, like the human pancreatic carcinoma cell line MIA PaCa-2 or MCF-7. Doxorubicins antiproliferative action on MIA PaCa-2 after 72 h incubation with the drug was as expected very strong (IC50 = 0.80 ± 0.13 μM), whereas octreotide was not able to inhibit cell growth by 50% up to a concentration of 150 μM. The cytotoxic effects of the conjugate expressed, as half maximal inhibitory concentration was much stronger compared to the precursor peptide, but, nevertheless, lower than that of doxorubicin (IC50 = 31.50 ± 1.74 μM). This characteristic feature of 12 is attributed to the different cellular uptake mechanisms of the substances. Doxorubicin is taken up quickly by passive diffusion, while octreotide enters cells by receptor-mediated endocytosis. Furthermore, it needs to be considered that the conjugate releases after cleavage by glutathione a doxorubicin derivative, which is still carrying a small cross-linker residue. Previous studies have shown that these types of molecules are, nevertheless, capable of successfully interacting with DNA, to mediate their cytotoxic properties, but to a lesser extent.
Click to Show/Hide
|
||||
| In Vitro Model | Pancreatic ductal adenocarcinoma | Capan-1 cell | CVCL_0237 | ||
References
