Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_02088
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
Octreotide doxorubicin conjugate
|
|||||
| PDC Status |
Investigative
|
|||||
| Indication |
In total 3 Indication(s)
|
|||||
| Structure |
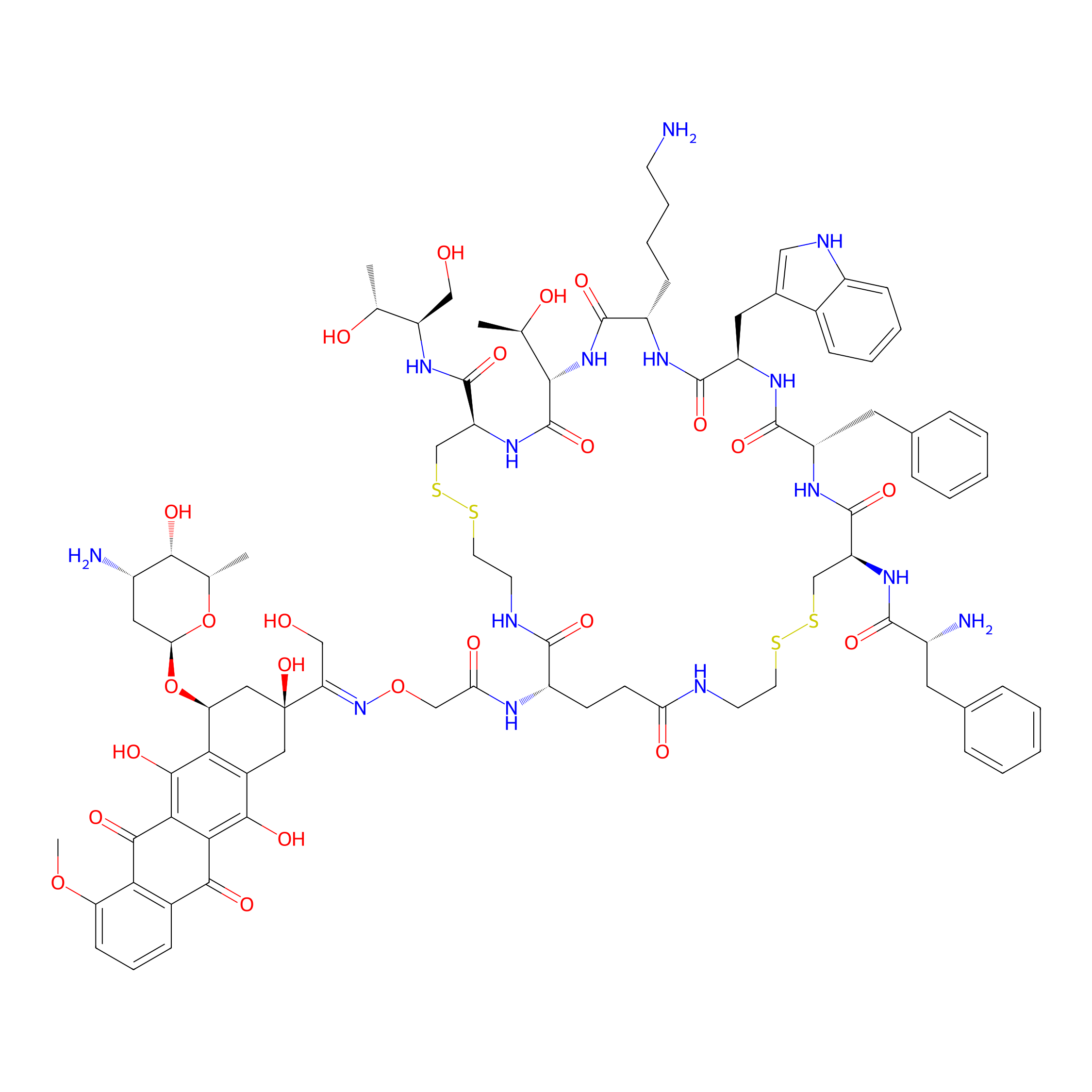
|
|||||
| Peptide Name |
Octreotide
|
Peptide Info | ||||
| Receptor Name |
Somatostatin receptor type 1 (SSTR1)
|
Receptor Info | ||||
| Drug Name |
Doxorubicin
|
Drug Info | ||||
| Therapeutic Target |
DNA topoisomerase 2-alpha (TOP2A)
|
Target Info | ||||
| Linker Name |
Aminooxyacetic acid
|
Linker Info | ||||
| Formula |
C87H113N15O24S4
|
|||||
| #Ro5 Violations (Lipinski): 4 | Molecular Weight | 1881.21 | ||||
| Lipid-water partition coefficient (xlogp) | -1.1082 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 22 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 32 | |||||
| Rotatable Bond Count (rotbonds) | 26 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
27.14 ± 2.47 μM
|
|||
| Administration Time | 72 h | ||||
| Evaluation Method | CellTiter-Glo cell proliferation assay | ||||
| MOA of PDC |
In the present work, we introduce a new approach to overcome all the aforementioned limitations. The cytotoxic drug doxorubicin is coupled to the tumor-targeting vector octreotide via a disulfide-intercalating cross-linking reagent. On the one hand, this reagent creates an oxime bond with the drug, and on the other hand, two disulfides with octreotide to keep the cyclic structure of the peptide. The combination of a hydrolytically stable oxime bond and disulfides leads to the formation of a novel bioconjugate superior to any previous anticancer drug-somatostatin analog hybrid as it allows the efficient release of the toxic cargo within the reducing environment of cancer cells. The versatility of the linker molecule described here will enable its future application not only in targeted drug delivery, but also in the chemical modification of therapeutic proteins.
Click to Show/Hide
|
||||
| Description |
We selected cells, where doxorubicin is typically applied and which overexpress somatostatin receptors, like the human pancreatic carcinoma cell line MIA PaCa-2 or MCF-7. Doxorubicins antiproliferative action on MIA PaCa-2 after 72 h incubation with the drug was as expected very strong (IC50 = 0.80 ± 0.13 μM), whereas octreotide was not able to inhibit cell growth by 50% up to a concentration of 150 μM. The cytotoxic effects of the conjugate expressed, as half maximal inhibitory concentration was much stronger compared to the precursor peptide, but, nevertheless, lower than that of doxorubicin (IC50 = 31.50 ± 1.74 μM). This characteristic feature of 12 is attributed to the different cellular uptake mechanisms of the substances. Doxorubicin is taken up quickly by passive diffusion, while octreotide enters cells by receptor-mediated endocytosis. Furthermore, it needs to be considered that the conjugate releases after cleavage by glutathione a doxorubicin derivative, which is still carrying a small cross-linker residue. Previous studies have shown that these types of molecules are, nevertheless, capable of successfully interacting with DNA, to mediate their cytotoxic properties, but to a lesser extent.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Pancreatic carcinoma | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
31.50 ± 1.74 μM
|
|||
| Administration Time | 72 h | ||||
| Evaluation Method | CellTiter-Glo cell proliferation assay | ||||
| MOA of PDC |
In the present work, we introduce a new approach to overcome all the aforementioned limitations. The cytotoxic drug doxorubicin is coupled to the tumor-targeting vector octreotide via a disulfide-intercalating cross-linking reagent. On the one hand, this reagent creates an oxime bond with the drug, and on the other hand, two disulfides with octreotide to keep the cyclic structure of the peptide. The combination of a hydrolytically stable oxime bond and disulfides leads to the formation of a novel bioconjugate superior to any previous anticancer drug-somatostatin analog hybrid as it allows the efficient release of the toxic cargo within the reducing environment of cancer cells. The versatility of the linker molecule described here will enable its future application not only in targeted drug delivery, but also in the chemical modification of therapeutic proteins.
Click to Show/Hide
|
||||
| Description |
We selected cells, where doxorubicin is typically applied and which overexpress somatostatin receptors, like the human pancreatic carcinoma cell line MIA PaCa-2 or MCF-7. Doxorubicins antiproliferative action on MIA PaCa-2 after 72 h incubation with the drug was as expected very strong (IC50 = 0.80 ± 0.13 μM), whereas octreotide was not able to inhibit cell growth by 50% up to a concentration of 150 μM. The cytotoxic effects of the conjugate expressed, as half maximal inhibitory concentration was much stronger compared to the precursor peptide, but, nevertheless, lower than that of doxorubicin (IC50 = 31.50 ± 1.74 μM). This characteristic feature of 12 is attributed to the different cellular uptake mechanisms of the substances. Doxorubicin is taken up quickly by passive diffusion, while octreotide enters cells by receptor-mediated endocytosis. Furthermore, it needs to be considered that the conjugate releases after cleavage by glutathione a doxorubicin derivative, which is still carrying a small cross-linker residue. Previous studies have shown that these types of molecules are, nevertheless, capable of successfully interacting with DNA, to mediate their cytotoxic properties, but to a lesser extent.
Click to Show/Hide
|
||||
| In Vitro Model | Pancreatic ductal adenocarcinoma | MIA PaCa-2 cell | CVCL_0428 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Pancreatic adenocarcinoma | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
48.90 ± 5.40 μM
|
|||
| Administration Time | 72 h | ||||
| Evaluation Method | CellTiter-Glo cell proliferation assay | ||||
| MOA of PDC |
In the present work, we introduce a new approach to overcome all the aforementioned limitations. The cytotoxic drug doxorubicin is coupled to the tumor-targeting vector octreotide via a disulfide-intercalating cross-linking reagent. On the one hand, this reagent creates an oxime bond with the drug, and on the other hand, two disulfides with octreotide to keep the cyclic structure of the peptide. The combination of a hydrolytically stable oxime bond and disulfides leads to the formation of a novel bioconjugate superior to any previous anticancer drug-somatostatin analog hybrid as it allows the efficient release of the toxic cargo within the reducing environment of cancer cells. The versatility of the linker molecule described here will enable its future application not only in targeted drug delivery, but also in the chemical modification of therapeutic proteins.
Click to Show/Hide
|
||||
| Description |
We selected cells, where doxorubicin is typically applied and which overexpress somatostatin receptors, like the human pancreatic carcinoma cell line MIA PaCa-2 or MCF-7. Doxorubicins antiproliferative action on MIA PaCa-2 after 72 h incubation with the drug was as expected very strong (IC50 = 0.80 ± 0.13 μM), whereas octreotide was not able to inhibit cell growth by 50% up to a concentration of 150 μM. The cytotoxic effects of the conjugate expressed, as half maximal inhibitory concentration was much stronger compared to the precursor peptide, but, nevertheless, lower than that of doxorubicin (IC50 = 31.50 ± 1.74 μM). This characteristic feature of 12 is attributed to the different cellular uptake mechanisms of the substances. Doxorubicin is taken up quickly by passive diffusion, while octreotide enters cells by receptor-mediated endocytosis. Furthermore, it needs to be considered that the conjugate releases after cleavage by glutathione a doxorubicin derivative, which is still carrying a small cross-linker residue. Previous studies have shown that these types of molecules are, nevertheless, capable of successfully interacting with DNA, to mediate their cytotoxic properties, but to a lesser extent.
Click to Show/Hide
|
||||
| In Vitro Model | Pancreatic ductal adenocarcinoma | Capan-1 cell | CVCL_0237 | ||
References
