Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_00019
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
[d-Cys6-des-Gly10-Pro9-α-azaGly-NH2]-GnRH-Dox
|
|||||
| PDC Status |
Investigative
|
|||||
| Indication |
In total 1 Indication(s)
|
|||||
| Structure |
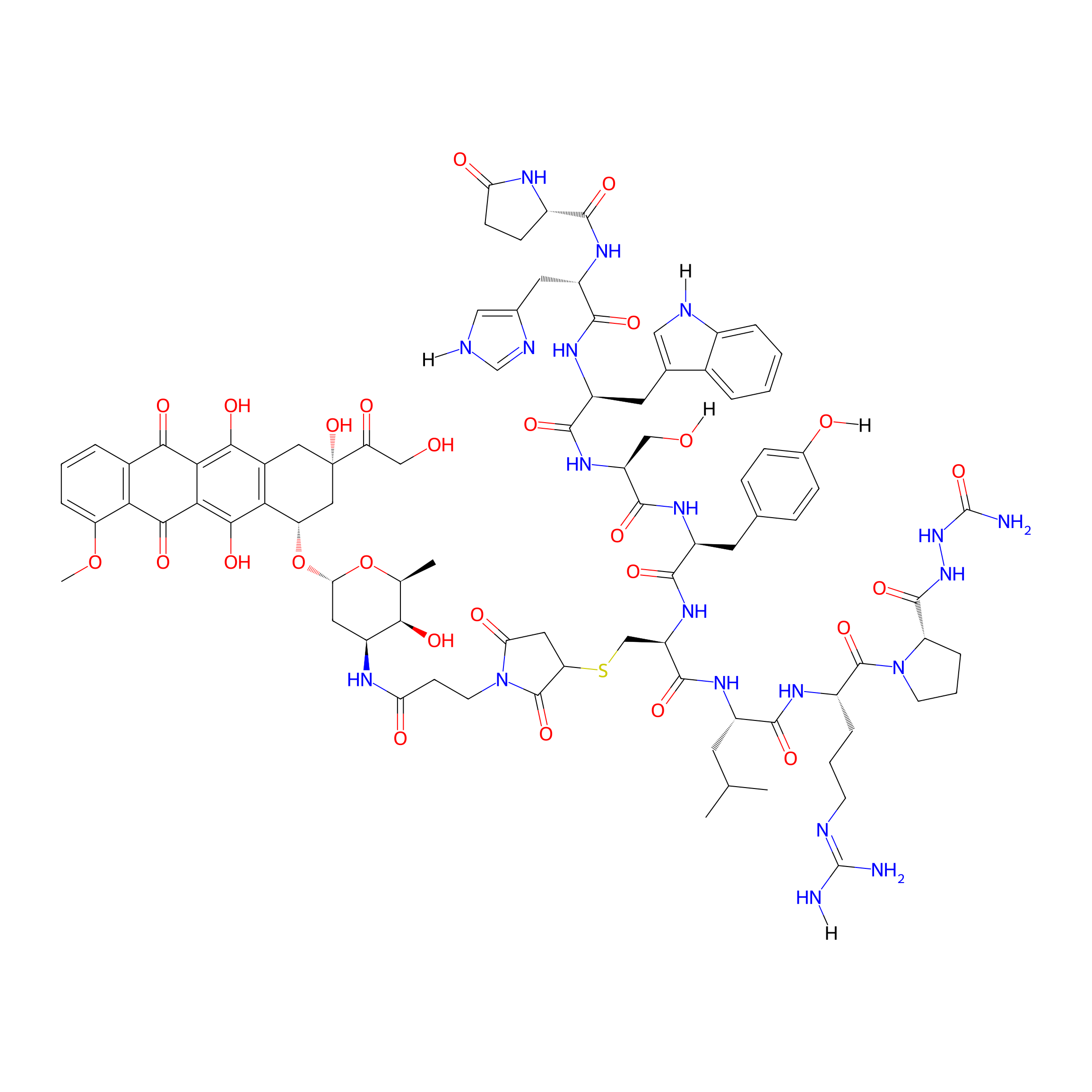
|
|||||
| Peptide Name |
GnRH analogs 3
|
Peptide Info | ||||
| Receptor Name |
Gonadotropin-releasing hormone receptor (GNRHR)
|
Receptor Info | ||||
| Drug Name |
Doxorubicin
|
Drug Info | ||||
| Therapeutic Target |
DNA topoisomerase 2-alpha (TOP2A)
|
Target Info | ||||
| Linker Name |
N-succinimidyl-3-maleimidopropionate
|
Linker Info | ||||
| Peptide Modified Type |
Amino acid modifications; The modification of binding with chemical macromolecules
|
|||||
| Modified Segment |
d-Cys6-des-Gly10-Pro9-α-azaGly-NH2
|
|||||
| Ternimal Modification |
C-terminal modification
|
|||||
| Formula |
C89H110N20O27S
|
|||||
| #Ro5 Violations (Lipinski): 5 | Molecular Weight | 1924.039 | ||||
| Lipid-water partition coefficient (xlogp) | -4.4976 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 23 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 30 | |||||
| Rotatable Bond Count (rotbonds) | 43 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Cell viability |
0.3
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 100 µM | ||||
| Evaluation Method | MTT assay | ||||
| Description |
The results showed that all the conjugates had lower antiproliferative effects than Dox at the tested concentrations (Figure 2A); this may be related to inefficient release of Dox from the conjugate caused by the relative stable thioether bond linkage between Dox-SMP and GnRH analog. The antiproliferative effects of the three conjugates were close at 25, 50, and 75 uM. While at 100 uM, conjugate II exhibited higher inhibitory effect than that of I and III.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Half life period | 5.23 h | ||||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Cell viability |
0.38
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 75 µM | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Vitamin C as a water-soluble vitamin is the reduced form of ascorbic acid. No significant adverse effect of taking high doses of vitamin C (over 2000 mg/day) has been reported due to the water-soluble feature of vitamin C. Vitamin C directly reacts with hydroxy, alkoxyl, and lipid peroxyl radicals and converts them to alcohol, water, and hydroperoxide lipid, respectively. It has been shown that taking vitamin C before radioiodine therapy can ameliorate the oxidative stress effect of radioiodine. The radioprotective effects of vitamin C are mainly due to its free radical scavenging activity.
Click to Show/Hide
|
||||
| Description |
The results showed that all the conjugates had lower antiproliferative effects than Dox at the tested concentrations (Figure 2A); this may be related to inefficient release of Dox from the conjugate caused by the relative stable thioether bond linkage between Dox-SMP and GnRH analog. The antiproliferative effects of the three conjugates were close at 25, 50, and 75 uM. While at 100 uM, conjugate II exhibited higher inhibitory effect than that of I and III.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Half life period | 5.23 h | ||||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Cell viability |
0.5
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 50 µM | ||||
| Evaluation Method | MTT assay | ||||
| Description |
The results showed that all the conjugates had lower antiproliferative effects than Dox at the tested concentrations (Figure 2A); this may be related to inefficient release of Dox from the conjugate caused by the relative stable thioether bond linkage between Dox-SMP and GnRH analog. The antiproliferative effects of the three conjugates were close at 25, 50, and 75 uM. While at 100 uM, conjugate II exhibited higher inhibitory effect than that of I and III.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Half life period | 5.23 h | ||||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Tumor | ||||
| Efficacy Data | Cell viability |
0.59
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 25 µM | ||||
| Evaluation Method | MTT assay | ||||
| Description |
The results showed that all the conjugates had lower antiproliferative effects than Dox at the tested concentrations (Figure 2A); this may be related to inefficient release of Dox from the conjugate caused by the relative stable thioether bond linkage between Dox-SMP and GnRH analog. The antiproliferative effects of the three conjugates were close at 25, 50, and 75 uM. While at 100 uM, conjugate II exhibited higher inhibitory effect than that of I and III.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
| Half life period | 5.23 h | ||||
References
