Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_00057
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
AEZS-108
|
|||||
| Synonyms |
Zoptarelin doxorubicin; 139570-93-7; AEZS-108; Zoptarelin doxorubicin [INN]; Zoptarelin doxorubicin [USAN]; AN-152; ZEN-008; 27844X2J29; Lys(6)-LHRH-doxorubicin; Zoptrex; AEZS 108; D-81858; DTXSID50161110; AN152; UNII-27844X2J29; Zoptarelin doxorubicin [USAN:INN]; AN 152; ZOPTARELIN DOXORUBICIN [WHO-DD]; HY-16532; CS-0006380; Q4651377
Click to Show/Hide
|
|||||
| PDC Status |
Terminated in Phase 3
|
|||||
| Indication |
In total 2 Indication(s)
|
|||||
| Structure |
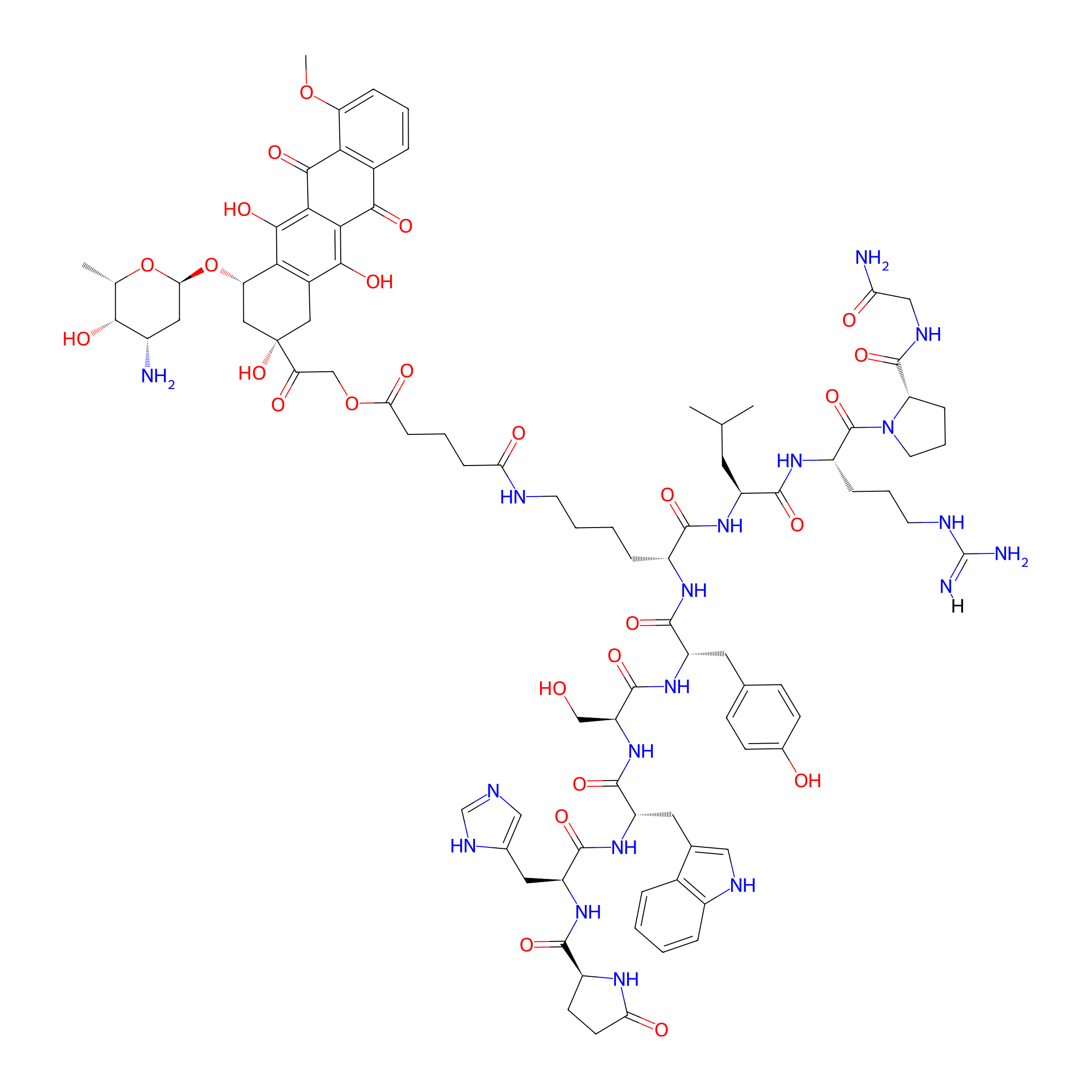
|
|||||
| Peptide Name |
[D-Lys6]-LH-RH
|
Peptide Info | ||||
| Receptor Name |
Gonadotropin-releasing hormone receptor (GNRHR)
|
Receptor Info | ||||
| Drug Name |
Doxorubicin
|
Drug Info | ||||
| Therapeutic Target |
DNA topoisomerase 2-alpha (TOP2A)
|
Target Info | ||||
| Linker Name |
Glutaric acid
|
Linker Info | ||||
| Peptide Modified Type |
Amino acid modifications
|
|||||
| Modified Segment |
Use D-amino acids instead of L-amino acids
|
|||||
| Drugbank ID | ||||||
| DrugMap ID | ||||||
| TTD ID | ||||||
| ChEBI ID | ||||||
| Formula |
C91H117N19O26
|
|||||
| #Ro5 Violations (Lipinski): 4 | Molecular Weight | 1893 | ||||
| Lipid-water partition coefficient (xlogp) | -0.4 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 23 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 29 | |||||
| Rotatable Bond Count (rotbonds) | 48 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Identified from the Human Clinical Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Castration and taxane resistant prostate cancer | ||||
| Efficacy Data | Alopecia toxicity |
52.00%
|
|||
| Description |
The most common all-grade adverse events were hematologic (22; 88%), fatigue (19; 76%), anorexia (13; 52%),alopecia(13; 52%), nausea (13; 52%), and vomiting (8;32%).
|
||||
| In Vivo Model | Men with histologically confirmed prostatic adenocarcinoma. | ||||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Castration and taxane resistant prostate cancer | ||||
| Efficacy Data | Fatigue |
76%
|
|||
| Description |
The most common all-grade adverse events were hematologic (22; 88%), fatigue (19; 76%), anorexia (13; 52%),alopecia(13; 52%), nausea (13; 52%), and vomiting (8;32%).
|
||||
| In Vivo Model | Men with histologically confirmed prostatic adenocarcinoma. | ||||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Castration and taxane resistant prostate cancer | ||||
| Efficacy Data | Grade ≥ 3 hematologic toxicity |
56%
|
|||
| Description |
Fourteen of 25 patients (56%) experienced a Grade ≥ 3 hematologic toxicity
|
||||
| In Vivo Model | Men with histologically confirmed prostatic adenocarcinoma. | ||||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Castration and taxane resistant prostate cancer | ||||
| Efficacy Data | Grade 3/4 nonhematologic toxicities |
24%
|
|||
| Description |
Six of 25 patients (24%) experienced Grade 3 or 4 nonhematologic toxicities
|
||||
| In Vivo Model | Men with histologically confirmed prostatic adenocarcinoma. | ||||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Castration and taxane resistant prostate cancer | ||||
| Efficacy Data | Hematologic toxicity |
88%
|
|||
| Description |
The most common all-grade adverse events were hematologic (22; 88%), fatigue (19; 76%), anorexia (13; 52%),alopecia(13; 52%), nausea (13; 52%), and vomiting (8;32%).
|
||||
| In Vivo Model | Men with histologically confirmed prostatic adenocarcinoma. | ||||
| Experiment 6 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Castration and taxane resistant prostate cancer | ||||
| Efficacy Data | Median overall survival (mOS) |
6.0 months
|
|||
| Description |
With a median follow-up of 16.1 months (range, 3.2-36.1), the median PFS was 3.8 months (95% confidence interval [CI], 2.1-4.4) and median OS was 6.0 months (95% CI, 4.2-10.1; Figure 3).
|
||||
| In Vivo Model | Men with histologically confirmed prostatic adenocarcinoma. | ||||
| Experiment 7 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Castration and taxane resistant prostate cancer | ||||
| Efficacy Data | Median progression-free survival (mPFS) |
3.8 months
|
|||
| Description |
With a median follow-up of 16.1 months (range, 3.2-36.1), the median PFS was 3.8 months (95% confidence interval [CI], 2.1-4.4) and median OS was 6.0 months (95% CI, 4.2-10.1; Figure 3).
|
||||
| In Vivo Model | Men with histologically confirmed prostatic adenocarcinoma. | ||||
| Experiment 8 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Castration and taxane resistant prostate cancer | ||||
| Efficacy Data | Nausea toxicity |
52%
|
|||
| Description |
The most common all-grade adverse events were hematologic (22; 88%), fatigue (19; 76%), anorexia (13; 52%),alopecia(13; 52%), nausea (13; 52%), and vomiting (8;32%).
|
||||
| In Vivo Model | Men with histologically confirmed prostatic adenocarcinoma. | ||||
| Experiment 9 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Castration and taxane resistant prostate cancer | ||||
| Efficacy Data | Neutropenia |
56%
|
|||
| Description |
The most common hematologic adverse event was neutropenia at 56% (all grades).
|
||||
| In Vivo Model | Men with histologically confirmed prostatic adenocarcinoma. | ||||
| Experiment 10 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Castration and taxane resistant prostate cancer | ||||
| Efficacy Data | Vomit |
32%
|
|||
| Description |
The most common all-grade adverse events were hematologic (22; 88%), fatigue (19; 76%), anorexia (13; 52%),alopecia(13; 52%), nausea (13; 52%), and vomiting (8;32%).
|
||||
| In Vivo Model | Men with histologically confirmed prostatic adenocarcinoma. | ||||
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Uveal melanoma | ||||
| Efficacy Data | Cell viability |
0%
|
|||
| Administration Time | 72 h | ||||
| Administration Dosage | 5 µM | ||||
| Evaluation Method | MTT assay | ||||
| Description |
OCM3DOX320cells did not show significant difference in cell viability in the presence of 1 uM DOX when compared to untreated control cells. 1 uM DOX induced significant cell death of OCM3 cells but did not cause significant cell death in OCM3DOX320cells, confirming DOX resistance. AN-152 at lower concentration (40 nM, 320 nM) increased cell proliferation significantly compared to equimolar dose of DOX which does not have any effect on cell viability at this concentration in OCM3 cells. However, higher concentrations of AN-152 can effectively inhibit cell proliferation in both cell lines. Higher concentrations (1-5 uM) of DOX and AN-152 showed no significantly different effect either on OCM3 or OCM3DOX320cells (Fig. 3).
Click to Show/Hide
|
||||
| In Vitro Model | Cutaneous melanoma | OCM-3 cell | CVCL_6937 | ||
| Half life period | 2 h | ||||
| Experiment 2 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Uveal melanoma | ||||
| Efficacy Data | Cell viability |
5%
|
|||
| Administration Time | 72 h | ||||
| Administration Dosage | 1 µM | ||||
| Evaluation Method | MTT assay | ||||
| Description |
OCM3DOX320cells did not show significant difference in cell viability in the presence of 1 uM DOX when compared to untreated control cells. 1 uM DOX induced significant cell death of OCM3 cells but did not cause significant cell death in OCM3DOX320cells, confirming DOX resistance. AN-152 at lower concentration (40 nM, 320 nM) increased cell proliferation significantly compared to equimolar dose of DOX which does not have any effect on cell viability at this concentration in OCM3 cells. However, higher concentrations of AN-152 can effectively inhibit cell proliferation in both cell lines. Higher concentrations (1-5 uM) of DOX and AN-152 showed no significantly different effect either on OCM3 or OCM3DOX320cells (Fig. 3).
Click to Show/Hide
|
||||
| In Vitro Model | Cutaneous melanoma | OCM-3 cell | CVCL_6937 | ||
| Half life period | 2 h | ||||
| Experiment 3 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Uveal melanoma | ||||
| Efficacy Data | Cell viability |
6%
|
|||
| Administration Time | 72 h | ||||
| Administration Dosage | 2.5 µM | ||||
| Evaluation Method | MTT assay | ||||
| Description |
OCM3DOX320cells did not show significant difference in cell viability in the presence of 1 uM DOX when compared to untreated control cells. 1 uM DOX induced significant cell death of OCM3 cells but did not cause significant cell death in OCM3DOX320cells, confirming DOX resistance. AN-152 at lower concentration (40 nM, 320 nM) increased cell proliferation significantly compared to equimolar dose of DOX which does not have any effect on cell viability at this concentration in OCM3 cells. However, higher concentrations of AN-152 can effectively inhibit cell proliferation in both cell lines. Higher concentrations (1-5 uM) of DOX and AN-152 showed no significantly different effect either on OCM3 or OCM3DOX320cells (Fig. 3).
Click to Show/Hide
|
||||
| In Vitro Model | Cutaneous melanoma | OCM-3 cell | CVCL_6937 | ||
| Half life period | 2 h | ||||
| Experiment 4 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Uveal melanoma | ||||
| Efficacy Data | Cell viability |
25%
|
|||
| Administration Time | 72 h | ||||
| Administration Dosage | 5 µM | ||||
| Evaluation Method | MTT assay | ||||
| Description |
OCM3DOX320cells did not show significant difference in cell viability in the presence of 1 uM DOX when compared to untreated control cells. 1 uM DOX induced significant cell death of OCM3 cells but did not cause significant cell death in OCM3DOX320cells, confirming DOX resistance. AN-152 at lower concentration (40 nM, 320 nM) increased cell proliferation significantly compared to equimolar dose of DOX which does not have any effect on cell viability at this concentration in OCM3 cells. However, higher concentrations of AN-152 can effectively inhibit cell proliferation in both cell lines. Higher concentrations (1-5 uM) of DOX and AN-152 showed no significantly different effect either on OCM3 or OCM3DOX320cells (Fig. 3).
Click to Show/Hide
|
||||
| In Vitro Model | Cutaneous melanoma | OCM3DOX320 cell | CVCL_6937 | ||
| Half life period | 2 h | ||||
| Experiment 5 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Uveal melanoma | ||||
| Efficacy Data | Cell viability |
36.30%
|
|||
| Administration Time | 24 h | ||||
| Administration Dosage | 5 µM | ||||
| Evaluation Method | MTS assay | ||||
| MOA of PDC |
Our results show that AEZS-108 upregulates the expression of MASPIN/SERPINB5 tumor suppressor gene, which is downregulated in normal uvea and UM specimens independently from the LHRH receptor-ligand interaction. AEZS-108 also substantially downregulates hypoxia-inducible factor 1 alpha (HIF1A) expression.
|
||||
| Description |
In order to investigate whether AEZS-108 inhibits cell proliferation and its extent, OCM3 cells were treated either with 5 M AEZS-108 or equal amount of doxorubicin. MTS assay was performed after 24 and 48 hours of treatment. AEZS-108 and doxorubicin have been shown to reduce cell proliferation by 36.3% (p< 0.001) and 62.9% (p< 0.001) respectively after 24 hours, and by 84.7% (p< 0.001) and 89.7% (p< 0.001) respectively after 48 hours, (Figure 2).
Click to Show/Hide
|
||||
| In Vitro Model | Cutaneous melanoma | OCM-3 cell | CVCL_6937 | ||
| Experiment 6 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Uveal melanoma | ||||
| Efficacy Data | Cell viability |
48%
|
|||
| Administration Time | 72 h | ||||
| Administration Dosage | 2.5 µM | ||||
| Evaluation Method | MTT assay | ||||
| Description |
OCM3DOX320cells did not show significant difference in cell viability in the presence of 1 uM DOX when compared to untreated control cells. 1 uM DOX induced significant cell death of OCM3 cells but did not cause significant cell death in OCM3DOX320cells, confirming DOX resistance. AN-152 at lower concentration (40 nM, 320 nM) increased cell proliferation significantly compared to equimolar dose of DOX which does not have any effect on cell viability at this concentration in OCM3 cells. However, higher concentrations of AN-152 can effectively inhibit cell proliferation in both cell lines. Higher concentrations (1-5 uM) of DOX and AN-152 showed no significantly different effect either on OCM3 or OCM3DOX320cells (Fig. 3).
Click to Show/Hide
|
||||
| In Vitro Model | Cutaneous melanoma | OCM3DOX320 cell | CVCL_6937 | ||
| Half life period | 2 h | ||||
| Experiment 7 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Uveal melanoma | ||||
| Efficacy Data | Cell viability |
80%
|
|||
| Administration Time | 72 h | ||||
| Administration Dosage | 1 µM | ||||
| Evaluation Method | MTT assay | ||||
| Description |
OCM3DOX320cells did not show significant difference in cell viability in the presence of 1 uM DOX when compared to untreated control cells. 1 uM DOX induced significant cell death of OCM3 cells but did not cause significant cell death in OCM3DOX320cells, confirming DOX resistance. AN-152 at lower concentration (40 nM, 320 nM) increased cell proliferation significantly compared to equimolar dose of DOX which does not have any effect on cell viability at this concentration in OCM3 cells. However, higher concentrations of AN-152 can effectively inhibit cell proliferation in both cell lines. Higher concentrations (1-5 uM) of DOX and AN-152 showed no significantly different effect either on OCM3 or OCM3DOX320cells (Fig. 3).
Click to Show/Hide
|
||||
| In Vitro Model | Cutaneous melanoma | OCM3DOX320 cell | CVCL_6937 | ||
| Half life period | 2 h | ||||
| Experiment 8 Reporting the Activity Data of This PDC | [3] | ||||
| Indication | Uveal melanoma | ||||
| Efficacy Data | Cell viability |
84.70%
|
|||
| Administration Time | 48 h | ||||
| Administration Dosage | 5 µM | ||||
| Evaluation Method | MTS assay | ||||
| MOA of PDC |
Our results show that AEZS-108 upregulates the expression of MASPIN/SERPINB5 tumor suppressor gene, which is downregulated in normal uvea and UM specimens independently from the LHRH receptor-ligand interaction. AEZS-108 also substantially downregulates hypoxia-inducible factor 1 alpha (HIF1A) expression.
|
||||
| Description |
In order to investigate whether AEZS-108 inhibits cell proliferation and its extent, OCM3 cells were treated either with 5 M AEZS-108 or equal amount of doxorubicin. MTS assay was performed after 24 and 48 hours of treatment. AEZS-108 and doxorubicin have been shown to reduce cell proliferation by 36.3% (p< 0.001) and 62.9% (p< 0.001) respectively after 24 hours, and by 84.7% (p< 0.001) and 89.7% (p< 0.001) respectively after 48 hours, (Figure 2).
Click to Show/Hide
|
||||
| In Vitro Model | Cutaneous melanoma | OCM-3 cell | CVCL_6937 | ||
| Experiment 9 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Uveal melanoma | ||||
| Efficacy Data | Cell viability |
105%
|
|||
| Administration Time | 72 h | ||||
| Administration Dosage | 320 nM | ||||
| Evaluation Method | MTT assay | ||||
| Description |
OCM3DOX320cells did not show significant difference in cell viability in the presence of 1 uM DOX when compared to untreated control cells. 1 uM DOX induced significant cell death of OCM3 cells but did not cause significant cell death in OCM3DOX320cells, confirming DOX resistance. AN-152 at lower concentration (40 nM, 320 nM) increased cell proliferation significantly compared to equimolar dose of DOX which does not have any effect on cell viability at this concentration in OCM3 cells. However, higher concentrations of AN-152 can effectively inhibit cell proliferation in both cell lines. Higher concentrations (1-5 uM) of DOX and AN-152 showed no significantly different effect either on OCM3 or OCM3DOX320cells (Fig. 3).
Click to Show/Hide
|
||||
| In Vitro Model | Cutaneous melanoma | OCM3DOX320 cell | CVCL_6937 | ||
| Half life period | 2 h | ||||
| Experiment 10 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Uveal melanoma | ||||
| Efficacy Data | Cell viability |
120%
|
|||
| Administration Time | 72 h | ||||
| Administration Dosage | 40 nM | ||||
| Evaluation Method | MTT assay | ||||
| Description |
OCM3DOX320cells did not show significant difference in cell viability in the presence of 1 uM DOX when compared to untreated control cells. 1 uM DOX induced significant cell death of OCM3 cells but did not cause significant cell death in OCM3DOX320cells, confirming DOX resistance. AN-152 at lower concentration (40 nM, 320 nM) increased cell proliferation significantly compared to equimolar dose of DOX which does not have any effect on cell viability at this concentration in OCM3 cells. However, higher concentrations of AN-152 can effectively inhibit cell proliferation in both cell lines. Higher concentrations (1-5 uM) of DOX and AN-152 showed no significantly different effect either on OCM3 or OCM3DOX320cells (Fig. 3).
Click to Show/Hide
|
||||
| In Vitro Model | Cutaneous melanoma | OCM3DOX320 cell | CVCL_6937 | ||
| Half life period | 2 h | ||||
| Experiment 11 Reporting the Activity Data of This PDC | [2] | ||||
| Indication | Uveal melanoma | ||||
| Efficacy Data | Cell viability |
130%
|
|||
| Administration Time | 72 h | ||||
| Administration Dosage | 40 nM | ||||
| Evaluation Method | MTT assay | ||||
| Description |
OCM3DOX320cells did not show significant difference in cell viability in the presence of 1 uM DOX when compared to untreated control cells. 1 uM DOX induced significant cell death of OCM3 cells but did not cause significant cell death in OCM3DOX320cells, confirming DOX resistance. AN-152 at lower concentration (40 nM, 320 nM) increased cell proliferation significantly compared to equimolar dose of DOX which does not have any effect on cell viability at this concentration in OCM3 cells. However, higher concentrations of AN-152 can effectively inhibit cell proliferation in both cell lines. Higher concentrations (1-5 uM) of DOX and AN-152 showed no significantly different effect either on OCM3 or OCM3DOX320cells (Fig. 3).
Click to Show/Hide
|
||||
| In Vitro Model | Cutaneous melanoma | OCM-3 cell | CVCL_6937 | ||
| Half life period | 2 h | ||||
References
