Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_00068
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
BT5528
|
|||||
| Synonyms |
BT5528; BT-5528; CIT3DOS0DW; CHEMBL5314953; Bt 5528; 2648849-70-9
Click to Show/Hide
|
|||||
| PDC Status |
Phase 1/2
|
|||||
| Indication |
In total 1 Indication(s)
|
|||||
| Structure |
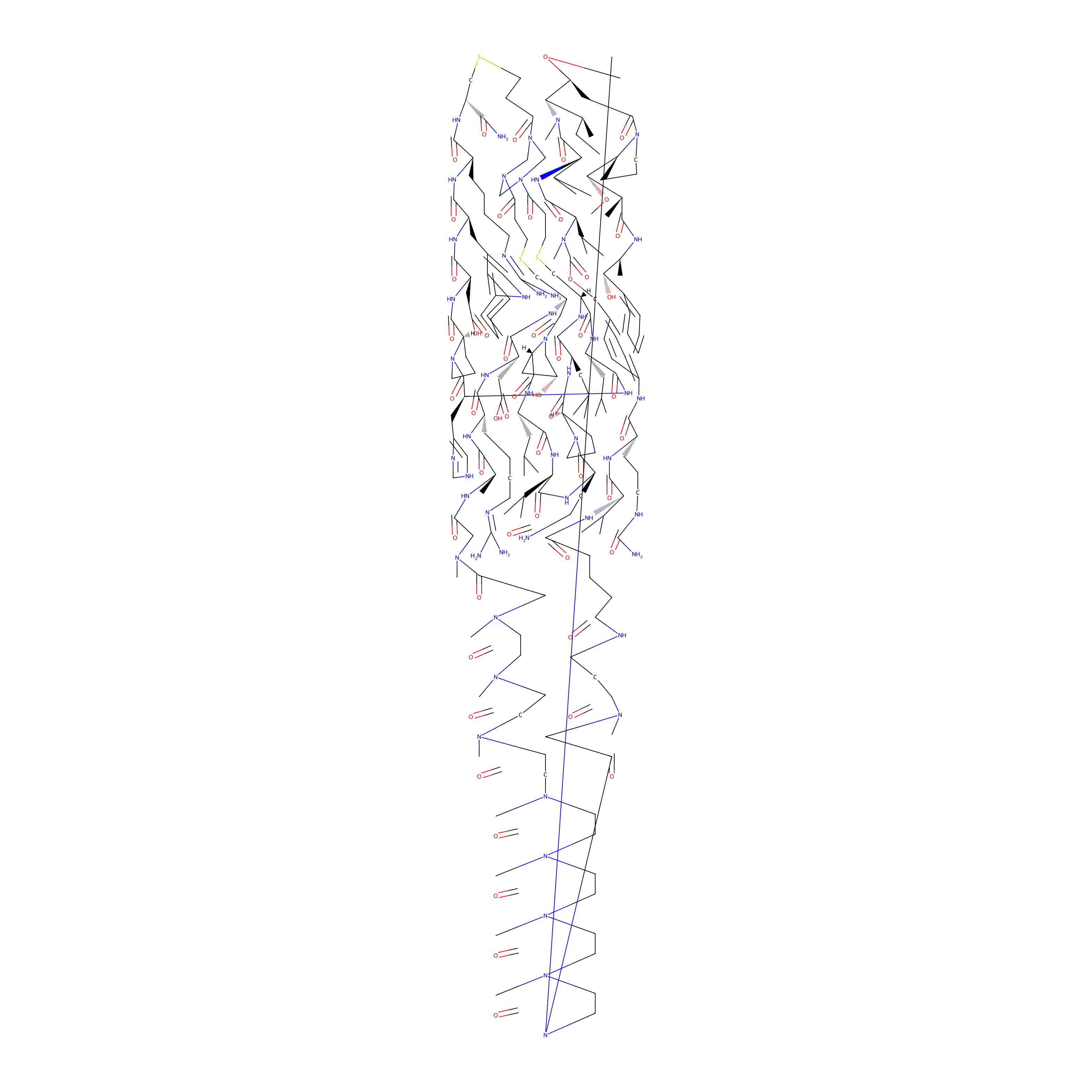
|
|||||
| Peptide Name |
BCY6099
|
Peptide Info | ||||
| Receptor Name |
Ephrin type-A receptor 2 (EPHA2)
|
Receptor Info | ||||
| Drug Name |
Monomethyl auristatin E
|
Drug Info | ||||
| Therapeutic Target |
Microtubule (MT)
|
Target Info | ||||
| Linker Name |
Val-Cit
|
Linker Info | ||||
| Peptide Modified Type |
Amino acid modifications; Cyclization modification
|
|||||
| Modified Segment |
Side-chain and side-chain cyclization; Use D-amino acids instead of L-amino acids
|
|||||
| Formula |
C201H315N53O52S3
|
|||||
| #Ro5 Violations (Lipinski): 5 | Molecular Weight | 4402 | ||||
| Lipid-water partition coefficient (xlogp) | -6.1 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 35 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 58 | |||||
| Rotatable Bond Count (rotbonds) | 95 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Discovered Using Cell Line-derived Xenograft Model
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Fibrosarcoma | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
50%
|
|||
| Administration Time | 20 day | ||||
| Administration Dosage | 0.167 mg/kg every week | ||||
| MOA of PDC |
Here we describe the development of BT5528, a bicyclic peptide (Bicycle) conjugated to the auristatin derivative maleimidocaproyl-monomethyl auristatin E to generate the Bicycle toxin conjugate BT5528. There are two potential mechanism of BT5528 bystander activity: extracellular linker cleavage and toxin penetration into neighboring cells, or receptor internalization and intracellular linker cleavage followed by release of cell penetrant toxin from lysed cells. The available data do not allow us to distinguish between these two mechanisms and it seems likely that BT5528 activity is mediated by toxin release following a combination of intracellular and extracellular linker cleavage.
Click to Show/Hide
|
||||
| Description |
BT5528 is efficacious in the PC3 xenograft model but control BTCs with noncleavable linkers and non-cell penetrant toxins lack comparable efficacy. A, The nonbinding BTC, BCY8245, is less active than BT5528 in the PC3 model (group mean ± SEM, n = 5) at both 0.5 and 0.0167 mg/kg dosing level (*, P < 0.05; **, P < 0.01; two-way ANOVA with Sidak's multiple comparisons test). B, Replacement of the cell penetrant toxin (MMAE: BT5528) with the non-cell penetrant toxin (MMAF: BCY10188) reduces activity in the PC3 model at 3, 1, and 0.33 mg/kg (group mean ± SEM; n = 5; *, P < 0.05; **, P < 0.01; two-way ANOVA with Sidak's multiple comparisons test). C, noncleavable linker chemistry abolishes activity in the HT1080 model; BCY6063, 10 mg/kg twice a week (group mean ± SEM, n = 3).
Click to Show/Hide
|
||||
| In Vivo Model | HT108 cells xenograft models in 6- to 8-week-old female balb/c nude or CB17-SCID mice. | ||||
| In Vitro Model | Fibrosarcoma | HT-1080 cell | CVCL_0317 | ||
| Half life period | 0.4 hour (Mouse); 0.3 hour (Rat); 0.6 hour (Nonhuman primate) | ||||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Fibrosarcoma | ||||
| Efficacy Data | Tumor Growth Inhibition value (TGI) |
87%
|
|||
| Administration Time | 20 day | ||||
| Administration Dosage | 0.5 mg/kg every week | ||||
| MOA of PDC |
Here we describe the development of BT5528, a bicyclic peptide (Bicycle) conjugated to the auristatin derivative maleimidocaproyl-monomethyl auristatin E to generate the Bicycle toxin conjugate BT5528. There are two potential mechanism of BT5528 bystander activity: extracellular linker cleavage and toxin penetration into neighboring cells, or receptor internalization and intracellular linker cleavage followed by release of cell penetrant toxin from lysed cells. The available data do not allow us to distinguish between these two mechanisms and it seems likely that BT5528 activity is mediated by toxin release following a combination of intracellular and extracellular linker cleavage.
Click to Show/Hide
|
||||
| Description |
BT5528 is efficacious in the PC3 xenograft model but control BTCs with noncleavable linkers and non-cell penetrant toxins lack comparable efficacy. A, The nonbinding BTC, BCY8245, is less active than BT5528 in the PC3 model (group mean ± SEM, n = 5) at both 0.5 and 0.0167 mg/kg dosing level (*, P < 0.05; **, P < 0.01; two-way ANOVA with Sidak's multiple comparisons test). B, Replacement of the cell penetrant toxin (MMAE: BT5528) with the non-cell penetrant toxin (MMAF: BCY10188) reduces activity in the PC3 model at 3, 1, and 0.33 mg/kg (group mean ± SEM; n = 5; *, P < 0.05; **, P < 0.01; two-way ANOVA with Sidak's multiple comparisons test). C, noncleavable linker chemistry abolishes activity in the HT1080 model; BCY6063, 10 mg/kg twice a week (group mean ± SEM, n = 3).
Click to Show/Hide
|
||||
| In Vivo Model | HT108 cells xenograft models in 6- to 8-week-old female balb/c nude or CB17-SCID mice. | ||||
| In Vitro Model | Fibrosarcoma | HT-1080 cell | CVCL_0317 | ||
| Half life period | 0.4 hour (Mouse); 0.3 hour (Rat); 0.6 hour (Nonhuman primate) | ||||
References
