Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_02128
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
Peptide 18-4 doxorubicin conjugate 2
|
|||||
| PDC Status |
Investigative
|
|||||
| Indication |
In total 5 Indication(s)
|
|||||
| Structure |
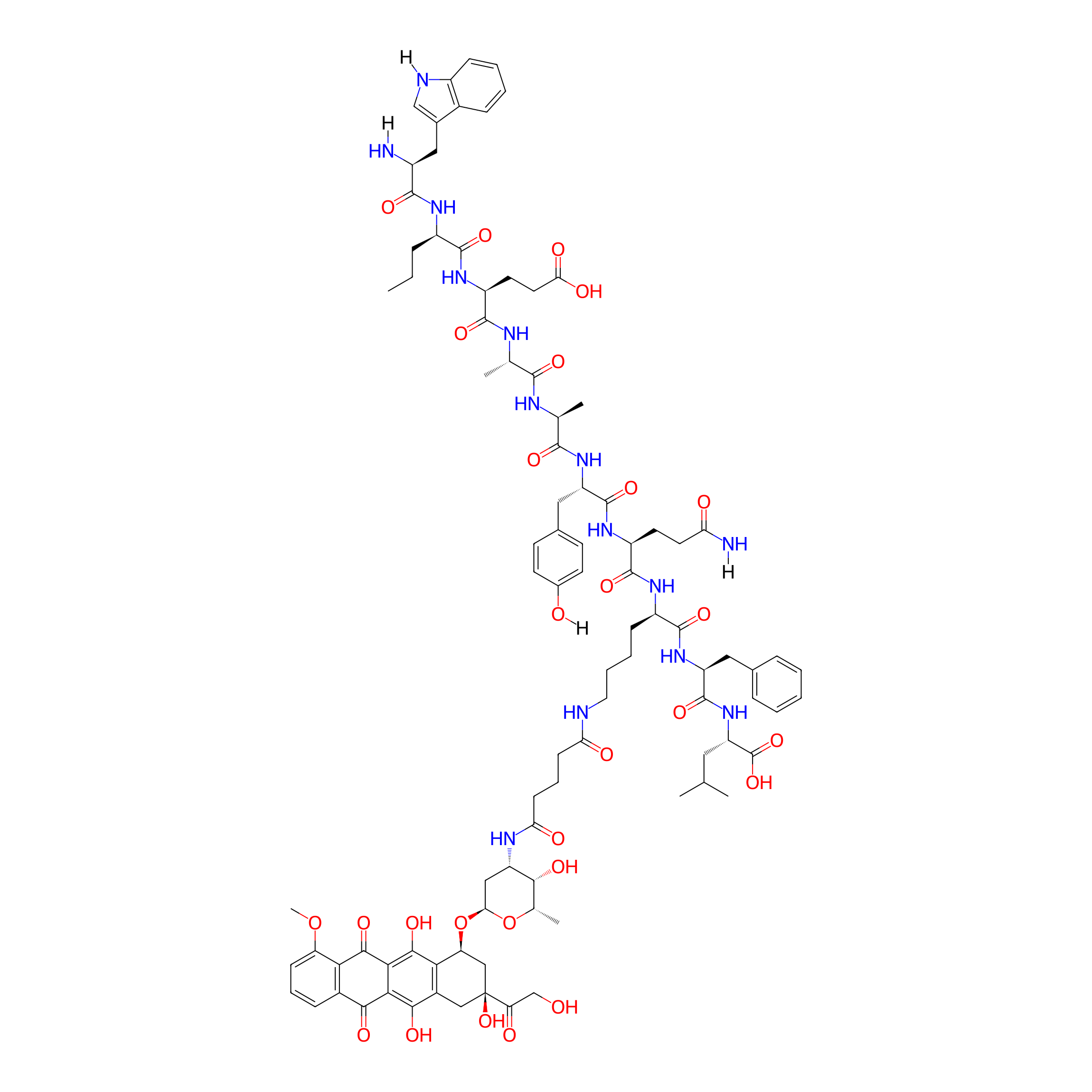
|
|||||
| Peptide Name |
Peptide 18-4
|
Peptide Info | ||||
| Receptor Name |
Keratin, type II cytoskeletal 1 (KRT1)
|
Receptor Info | ||||
| Drug Name |
Doxorubicin
|
Drug Info | ||||
| Therapeutic Target |
DNA topoisomerase 2-alpha (TOP2A)
|
Target Info | ||||
| Linker Name |
Glutaric acid
|
Linker Info | ||||
| Formula |
C94H120N14O28
|
|||||
| #Ro5 Violations (Lipinski): 4 | Molecular Weight | 1894.064 | ||||
| Lipid-water partition coefficient (xlogp) | 0.322 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 22 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 27 | |||||
| Rotatable Bond Count (rotbonds) | 52 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Amelanotic melanoma | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
18.6 ± 2.5 μM
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Here we report the design and synthesis of two new peptide-Dox conjugates (1 and 2) for the specific delivery of Dox to the breast cancer cells and the ability to overcome P-glycoprotein multidrug resistance pathway in both drug-sensitive and drug-resistant cancer cells. Peptide-Dox conjugates were evaluated for DOX release in human serum, intracellular delivery compared to free Dox in three cancerous cells (MCF-7, MDA-MB-435, and MDA-MB-435-MDR) and two noncancerous cell lines (HUVEC and MCF-10A), and cytotoxicity compared to free Dox. Results show that both the peptide-Dox conjugates (1 and 2) enter sensitive and resistant cell lines with minimal uptake in normal cells compared to free Dox. Cellular uptake is most likely mediated by a cell specific receptor, as the amount of internalized conjugates significantly decreased in the presence of excess free peptide. Importantly, conjugate 1 is equally cytotoxic as Dox in drug sensitive breast cancer cells and 4 times more potent than free Dox in Dox resistant cell line. Overall, the peptide-Dox ester conjugate 1 showed better breast targeting efficacy than the amide conjugate 2, most likely due to the slow release of Dox from the stable amide linkage.
Click to Show/Hide
|
||||
| Description |
The cytotoxicity experiment was done by incubating the cells with different treatments for 48 h. The results show that conjugate 1 is quite similar to free Dox for toxicity to MCF-7 and MDA-MB-435 cancer cells. In contrast, conjugate 2 was ?20 times less cytotoxic to breast cancerous cells compared to free Dox. This could be attributed to the stability of the amide conjugate 2 inside the cells. Furthermore, the cytotoxicity of conjugate 1 in doxorubicin-resistant cell model MDA-MB-435-MDR (IC50 = 5.4 μM) is 4 times more than free Dox (IC50 = 22 μM). In normal cells (HUVEC and MCF-10A) the two conjugates were 3540 times less toxic compared to breast cancer cells, whereas free Dox was equally cytotoxic (equal IC50) to breast cancer cells and noncancerous cells. Overall these results provide clear evidence that of the two conjugates (conjugate 1 and 2), conjugate 1 has optimal characteristics for specific Dox targeting to breast cancer cells.
Click to Show/Hide
|
||||
| In Vitro Model | Amelanotic melanoma | MDA-MB-435 cell | CVCL_0417 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Amelanotic melanoma | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
19.7 ± 3.1 μM
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Here we report the design and synthesis of two new peptide-Dox conjugates (1 and 2) for the specific delivery of Dox to the breast cancer cells and the ability to overcome P-glycoprotein multidrug resistance pathway in both drug-sensitive and drug-resistant cancer cells. Peptide-Dox conjugates were evaluated for DOX release in human serum, intracellular delivery compared to free Dox in three cancerous cells (MCF-7, MDA-MB-435, and MDA-MB-435-MDR) and two noncancerous cell lines (HUVEC and MCF-10A), and cytotoxicity compared to free Dox. Results show that both the peptide-Dox conjugates (1 and 2) enter sensitive and resistant cell lines with minimal uptake in normal cells compared to free Dox. Cellular uptake is most likely mediated by a cell specific receptor, as the amount of internalized conjugates significantly decreased in the presence of excess free peptide. Importantly, conjugate 1 is equally cytotoxic as Dox in drug sensitive breast cancer cells and 4 times more potent than free Dox in Dox resistant cell line. Overall, the peptide-Dox ester conjugate 1 showed better breast targeting efficacy than the amide conjugate 2, most likely due to the slow release of Dox from the stable amide linkage.
Click to Show/Hide
|
||||
| Description |
The cytotoxicity experiment was done by incubating the cells with different treatments for 48 h. The results show that conjugate 1 is quite similar to free Dox for toxicity to MCF-7 and MDA-MB-435 cancer cells. In contrast, conjugate 2 was ?20 times less cytotoxic to breast cancerous cells compared to free Dox. This could be attributed to the stability of the amide conjugate 2 inside the cells. Furthermore, the cytotoxicity of conjugate 1 in doxorubicin-resistant cell model MDA-MB-435-MDR (IC50 = 5.4 μM) is 4 times more than free Dox (IC50 = 22 μM). In normal cells (HUVEC and MCF-10A) the two conjugates were 3540 times less toxic compared to breast cancer cells, whereas free Dox was equally cytotoxic (equal IC50) to breast cancer cells and noncancerous cells. Overall these results provide clear evidence that of the two conjugates (conjugate 1 and 2), conjugate 1 has optimal characteristics for specific Dox targeting to breast cancer cells.
Click to Show/Hide
|
||||
| In Vitro Model | Amelanotic melanoma | MDA-MB-435 cell | CVCL_0417 | ||
| Experiment 3 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
40.5 ± 4.3 μM
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Here we report the design and synthesis of two new peptide-Dox conjugates (1 and 2) for the specific delivery of Dox to the breast cancer cells and the ability to overcome P-glycoprotein multidrug resistance pathway in both drug-sensitive and drug-resistant cancer cells. Peptide-Dox conjugates were evaluated for DOX release in human serum, intracellular delivery compared to free Dox in three cancerous cells (MCF-7, MDA-MB-435, and MDA-MB-435-MDR) and two noncancerous cell lines (HUVEC and MCF-10A), and cytotoxicity compared to free Dox. Results show that both the peptide-Dox conjugates (1 and 2) enter sensitive and resistant cell lines with minimal uptake in normal cells compared to free Dox. Cellular uptake is most likely mediated by a cell specific receptor, as the amount of internalized conjugates significantly decreased in the presence of excess free peptide. Importantly, conjugate 1 is equally cytotoxic as Dox in drug sensitive breast cancer cells and 4 times more potent than free Dox in Dox resistant cell line. Overall, the peptide-Dox ester conjugate 1 showed better breast targeting efficacy than the amide conjugate 2, most likely due to the slow release of Dox from the stable amide linkage.
Click to Show/Hide
|
||||
| Description |
The cytotoxicity experiment was done by incubating the cells with different treatments for 48 h. The results show that conjugate 1 is quite similar to free Dox for toxicity to MCF-7 and MDA-MB-435 cancer cells. In contrast, conjugate 2 was ?20 times less cytotoxic to breast cancerous cells compared to free Dox. This could be attributed to the stability of the amide conjugate 2 inside the cells. Furthermore, the cytotoxicity of conjugate 1 in doxorubicin-resistant cell model MDA-MB-435-MDR (IC50 = 5.4 μM) is 4 times more than free Dox (IC50 = 22 μM). In normal cells (HUVEC and MCF-10A) the two conjugates were 3540 times less toxic compared to breast cancer cells, whereas free Dox was equally cytotoxic (equal IC50) to breast cancer cells and noncancerous cells. Overall these results provide clear evidence that of the two conjugates (conjugate 1 and 2), conjugate 1 has optimal characteristics for specific Dox targeting to breast cancer cells.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | MCF-10A cell | CVCL_0598 | ||
| Experiment 4 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Solid tumor | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
50.9 ± 3.2 μM
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Here we report the design and synthesis of two new peptide-Dox conjugates (1 and 2) for the specific delivery of Dox to the breast cancer cells and the ability to overcome P-glycoprotein multidrug resistance pathway in both drug-sensitive and drug-resistant cancer cells. Peptide-Dox conjugates were evaluated for DOX release in human serum, intracellular delivery compared to free Dox in three cancerous cells (MCF-7, MDA-MB-435, and MDA-MB-435-MDR) and two noncancerous cell lines (HUVEC and MCF-10A), and cytotoxicity compared to free Dox. Results show that both the peptide-Dox conjugates (1 and 2) enter sensitive and resistant cell lines with minimal uptake in normal cells compared to free Dox. Cellular uptake is most likely mediated by a cell specific receptor, as the amount of internalized conjugates significantly decreased in the presence of excess free peptide. Importantly, conjugate 1 is equally cytotoxic as Dox in drug sensitive breast cancer cells and 4 times more potent than free Dox in Dox resistant cell line. Overall, the peptide-Dox ester conjugate 1 showed better breast targeting efficacy than the amide conjugate 2, most likely due to the slow release of Dox from the stable amide linkage.
Click to Show/Hide
|
||||
| Description |
The cytotoxicity experiment was done by incubating the cells with different treatments for 48 h. The results show that conjugate 1 is quite similar to free Dox for toxicity to MCF-7 and MDA-MB-435 cancer cells. In contrast, conjugate 2 was ?20 times less cytotoxic to breast cancerous cells compared to free Dox. This could be attributed to the stability of the amide conjugate 2 inside the cells. Furthermore, the cytotoxicity of conjugate 1 in doxorubicin-resistant cell model MDA-MB-435-MDR (IC50 = 5.4 μM) is 4 times more than free Dox (IC50 = 22 μM). In normal cells (HUVEC and MCF-10A) the two conjugates were 3540 times less toxic compared to breast cancer cells, whereas free Dox was equally cytotoxic (equal IC50) to breast cancer cells and noncancerous cells. Overall these results provide clear evidence that of the two conjugates (conjugate 1 and 2), conjugate 1 has optimal characteristics for specific Dox targeting to breast cancer cells.
Click to Show/Hide
|
||||
| In Vitro Model | Normal | Human umbilical vein endothelial cell | Homo sapiens | ||
| Experiment 5 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Breast cancer | ||||
| Efficacy Data | Half maximal inhibitory concentration (IC50) |
191 ± 2.8 μM
|
|||
| Administration Time | 48 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
Here we report the design and synthesis of two new peptide-Dox conjugates (1 and 2) for the specific delivery of Dox to the breast cancer cells and the ability to overcome P-glycoprotein multidrug resistance pathway in both drug-sensitive and drug-resistant cancer cells. Peptide-Dox conjugates were evaluated for DOX release in human serum, intracellular delivery compared to free Dox in three cancerous cells (MCF-7, MDA-MB-435, and MDA-MB-435-MDR) and two noncancerous cell lines (HUVEC and MCF-10A), and cytotoxicity compared to free Dox. Results show that both the peptide-Dox conjugates (1 and 2) enter sensitive and resistant cell lines with minimal uptake in normal cells compared to free Dox. Cellular uptake is most likely mediated by a cell specific receptor, as the amount of internalized conjugates significantly decreased in the presence of excess free peptide. Importantly, conjugate 1 is equally cytotoxic as Dox in drug sensitive breast cancer cells and 4 times more potent than free Dox in Dox resistant cell line. Overall, the peptide-Dox ester conjugate 1 showed better breast targeting efficacy than the amide conjugate 2, most likely due to the slow release of Dox from the stable amide linkage.
Click to Show/Hide
|
||||
| Description |
The cytotoxicity experiment was done by incubating the cells with different treatments for 48 h. The results show that conjugate 1 is quite similar to free Dox for toxicity to MCF-7 and MDA-MB-435 cancer cells. In contrast, conjugate 2 was ?20 times less cytotoxic to breast cancerous cells compared to free Dox. This could be attributed to the stability of the amide conjugate 2 inside the cells. Furthermore, the cytotoxicity of conjugate 1 in doxorubicin-resistant cell model MDA-MB-435-MDR (IC50 = 5.4 μM) is 4 times more than free Dox (IC50 = 22 μM). In normal cells (HUVEC and MCF-10A) the two conjugates were 3540 times less toxic compared to breast cancer cells, whereas free Dox was equally cytotoxic (equal IC50) to breast cancer cells and noncancerous cells. Overall these results provide clear evidence that of the two conjugates (conjugate 1 and 2), conjugate 1 has optimal characteristics for specific Dox targeting to breast cancer cells.
Click to Show/Hide
|
||||
| In Vitro Model | Invasive breast carcinoma | MCF-7 cell | CVCL_0031 | ||
References
