Peptide-drug Conjugate Information
General Information of This Peptide-drug Conjugate (PDC)
| PDC ID |
PDC_00081
|
|||||
|---|---|---|---|---|---|---|
| PDC Name |
CPP-SA-PTX
|
|||||
| Synonyms |
CPP-PTX conjugate; CPP-SA-PTX; GLXC-27449
Click to Show/Hide
|
|||||
| PDC Status |
Investigative
|
|||||
| Indication |
In total 1 Indication(s)
|
|||||
| Structure |
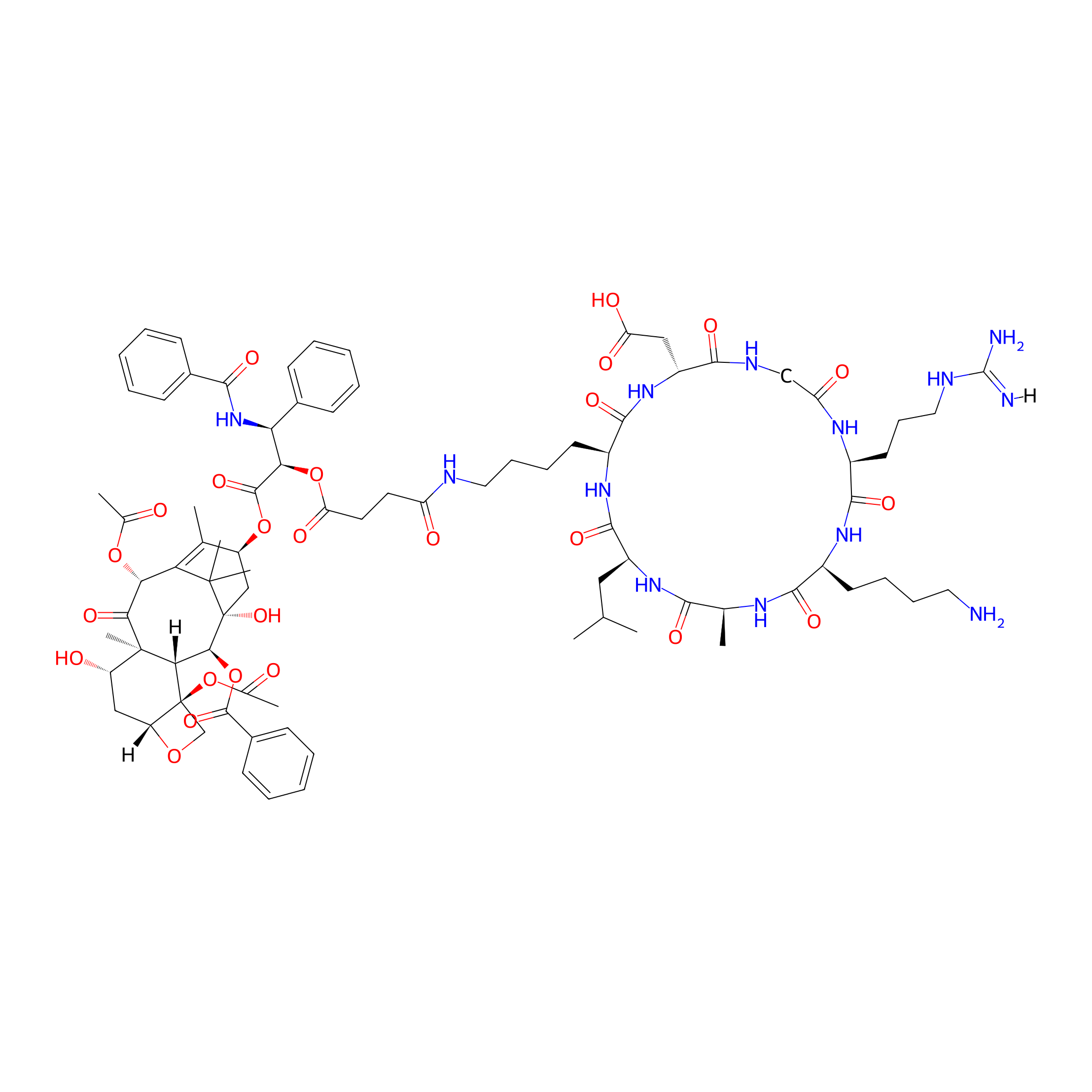
|
|||||
| Peptide Name |
RGDKLAK
|
Peptide Info | ||||
| Receptor Name |
Integrin alpha-V; Integrin beta-3 (ITGAV; ITGB3)
|
Receptor Info | ||||
| Drug Name |
Paclitaxel
|
Drug Info | ||||
| Therapeutic Target |
Microtubule (MT)
|
Target Info | ||||
| Linker Name |
Succinic Acid
|
Linker Info | ||||
| Peptide Modified Type |
Cyclization modification
|
|||||
| Modified Segment |
Head-to-tail cyclization
|
|||||
| Formula |
C84H113N13O25
|
|||||
| #Ro5 Violations (Lipinski): 5 | Molecular Weight | 1704.9 | ||||
| Lipid-water partition coefficient (xlogp) | -2.3 | |||||
| Hydrogen Bond Donor Count (hbonddonor) | 16 | |||||
| Hydrogen Bond Acceptor Count (hbondacc) | 27 | |||||
| Rotatable Bond Count (rotbonds) | 37 | |||||
Full List of Activity Data of This Peptide-drug Conjugate
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Malignant glioblastoma | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) |
20.55 ± 1.02 nM
|
|||
| Administration Time | 24 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
A highly sensitive, nontoxic, hydrophilic cell-penetrating peptide (CPP = c[RGDKLAK]) was selected for the construction of an effective peptide-drug conjugate (PDC). A hydrophobic drug paclitaxel (PTX) was successfully conjugated with CPP via ester linkage with succinic acid (SA) as a pH-cleavable linker moiety. The characterization techniques employed in this study indicate the >95% purity of the resulting PDC (CPP-SA-PTX). The in vitro studies show that our proposed PDC exhibits enhanced stability (˜90%) and cytotoxicity (EC50 = 8.32 ± 0.09 nM). Besides the excellent solubility of PDC in water, the PTX effect on positive β-tubulin-III indicates that the drug releases retained pharmacological properties. Additionally, in vivo, therapeutic-dose treatment reveals the prominent tumor-growth inhibitory effects (2.82-3.24-fold) of PDC in tumor mice models. Subsequently, these observations confirmed that our novel-designed PDC (CPP-SA-PTX) adduct may serve as a promising therapeutic agent to treat glioblastoma.
Click to Show/Hide
|
||||
| Description |
Furthermore, it was also observed that PDC shows time-dependent cytotoxicity against U87MG cells, as at 12 h drug treatment. PDC shows a slightly lower inhibitory effect on cell survival (EC50 = 25.82 ± 0.31 nM) than PTX alone (EC50 = 12.25 ± 0.13 nM), and CPP-SA alone shows higher EC50 = 54.37 ± 0.24 in U87MG cells. Besides this, it was previously observed that CPP-treated healthy VERO cells showed 88.05 ± 0.86% viable cells even using 10 uM concentration, indicating the specificity of CPP for glioblastoma cells. However, at 24 h incubation period, our PDC shows a significantly enhanced cell survival inhibitory effect with EC50 = 8.32 ± 0.09 nM, suggesting the delayed effect of PDC because of the time required for the intracellular pH to cause maximal cleavage of PTX from the PDC. Moreover, it also shows that CPP-SA has a lower cytotoxicity effect on glioblastoma cells compared with PTX alone and PDC, indicating its specificity as a carrier and selectivity for integrin receptors overexpressed on the cell surface. Additionally, we have also studied the potential of our novel-designed PDC to internalize into PTX-resistant glioblastoma (U87MG-PR) cells to show cytotoxic activity upon intracellular cleavage of PTX from CPP. The data presented in Figure 3b reveal approximately 15% viability increases in U87MG-PR cells compared with parent U87MG cells. It can be seen in the inset in Figure 3b that PTX alone (EC50 = 41.3 ± 1.5 nM) showed significantly 2-fold reduced cytotoxic activity in U87MG-PR cells in comparison with our novel-designed PDC having EC50 = 20.55 ± 1.02 nM. It was also observed in Figure 3b that at lower concentrations (0-10 nM) the viable cell count was ˜75%; however, it significantly decreased to ˜20% viability with an increase in concentration (40 nM) of the test samples, highlighting the dose-dependent cytotoxicity behavior of PDC in a 24 h incubation period.
Click to Show/Hide
|
||||
| In Vitro Model | Glioblastoma | U87MG-PR cell | CVCL_0022 | ||
| Experiment 2 Reporting the Activity Data of This PDC | [1] | ||||
| Indication | Malignant glioblastoma | ||||
| Efficacy Data | Half Maximal Effective Concentration (EC50) |
25.82 ± 0.31 nM
|
|||
| Administration Time | 24 h | ||||
| Evaluation Method | MTT assay | ||||
| MOA of PDC |
A highly sensitive, nontoxic, hydrophilic cell-penetrating peptide (CPP = c[RGDKLAK]) was selected for the construction of an effective peptide-drug conjugate (PDC). A hydrophobic drug paclitaxel (PTX) was successfully conjugated with CPP via ester linkage with succinic acid (SA) as a pH-cleavable linker moiety. The characterization techniques employed in this study indicate the >95% purity of the resulting PDC (CPP-SA-PTX). The in vitro studies show that our proposed PDC exhibits enhanced stability (˜90%) and cytotoxicity (EC50 = 8.32 ± 0.09 nM). Besides the excellent solubility of PDC in water, the PTX effect on positive β-tubulin-III indicates that the drug releases retained pharmacological properties. Additionally, in vivo, therapeutic-dose treatment reveals the prominent tumor-growth inhibitory effects (2.82-3.24-fold) of PDC in tumor mice models. Subsequently, these observations confirmed that our novel-designed PDC (CPP-SA-PTX) adduct may serve as a promising therapeutic agent to treat glioblastoma.
Click to Show/Hide
|
||||
| Description |
Furthermore, it was also observed that PDC shows time-dependent cytotoxicity against U87MG cells, as at 12 h drug treatment. PDC shows a slightly lower inhibitory effect on cell survival (EC50 = 25.82 ± 0.31 nM) than PTX alone (EC50 = 12.25 ± 0.13 nM), and CPP-SA alone shows higher EC50 = 54.37 ± 0.24 in U87MG cells. Besides this, it was previously observed that CPP-treated healthy VERO cells showed 88.05 ± 0.86% viable cells even using 10 uM concentration, indicating the specificity of CPP for glioblastoma cells. However, at 24 h incubation period, our PDC shows a significantly enhanced cell survival inhibitory effect with EC50 = 8.32 ± 0.09 nM, suggesting the delayed effect of PDC because of the time required for the intracellular pH to cause maximal cleavage of PTX from the PDC. Moreover, it also shows that CPP-SA has a lower cytotoxicity effect on glioblastoma cells compared with PTX alone and PDC, indicating its specificity as a carrier and selectivity for integrin receptors overexpressed on the cell surface. Additionally, we have also studied the potential of our novel-designed PDC to internalize into PTX-resistant glioblastoma (U87MG-PR) cells to show cytotoxic activity upon intracellular cleavage of PTX from CPP. The data presented in Figure 3b reveal approximately 15% viability increases in U87MG-PR cells compared with parent U87MG cells. It can be seen in the inset in Figure 3b that PTX alone (EC50 = 41.3 ± 1.5 nM) showed significantly 2-fold reduced cytotoxic activity in U87MG-PR cells in comparison with our novel-designed PDC having EC50 = 20.55 ± 1.02 nM. It was also observed in Figure 3b that at lower concentrations (0-10 nM) the viable cell count was ˜75%; however, it significantly decreased to ˜20% viability with an increase in concentration (40 nM) of the test samples, highlighting the dose-dependent cytotoxicity behavior of PDC in a 24 h incubation period.
Click to Show/Hide
|
||||
| In Vitro Model | Glioblastoma | U-87MG cell | CVCL_0022 | ||
References
